What is AP Biology Unit 1?
It covers water chemistry, biological elements, macromolecules, structure-function, and nucleic acids.
Most lost Unit 1 points come from mixing up cohesion vs adhesion, lipids as non-polymers, or DNA vs RNA details.
Unit 1 is the molecular foundation for every later AP Biology unit. Follow one path from water chemistry through macromolecules, then review and practice — at your own pace.
11 connected stops. Follow them in order or jump to where you need help.
Each stop has its own guide with flashcards and MCQs. Start anywhere.

Follow the learning path above, or filter topic guides below. Each deep dive has flashcards, MCQs, and structure-function practice.

Water's polarity and hydrogen bonding explain many properties that support life.
Open guide →CHNOPS elements build the molecules living systems need.
Open guide →Small building blocks combine into larger biological molecules.
Open guide →Cells build molecules by releasing water and break molecules by using water.
Open guide →The four major groups are carbohydrates, lipids, proteins, and nucleic acids.
Open guide →Sugars and polysaccharides support energy, storage, and structure.
Open guide →Hydrophobic molecules support long-term energy storage, membranes, and signaling.
Open guide →Amino acid chains fold into functional proteins such as enzymes and receptors.
Open guide →DNA and RNA store and help use genetic information.
Open guide →Cumulative Chemistry of Life review with charts, 15 mixed MCQs, mini FRQs, and weak-area links.
Open review →50 AP-style MCQs on this page with explanations, score tracking, and difficulty that builds through the set.
Start hub MCQs →Full Chemistry of Life MCQ and FRQ set in a quiz-first flow (dedicated practice page).
Open practice →Fast review answers for common Unit 1 questions.
It covers water chemistry, biological elements, macromolecules, structure-function, and nucleic acids.
About 8 to 11 percent directly, with concepts reused in later units and FRQs.
Explaining how molecular structure causes biological function in a specific scenario.
Start with properties of water and CHNOPS, then move to macromolecules and nucleic acids.
Quick concept anchors for daily review.
A weak attraction involving partial charges that helps explain cohesion, adhesion, and water's temperature behavior.
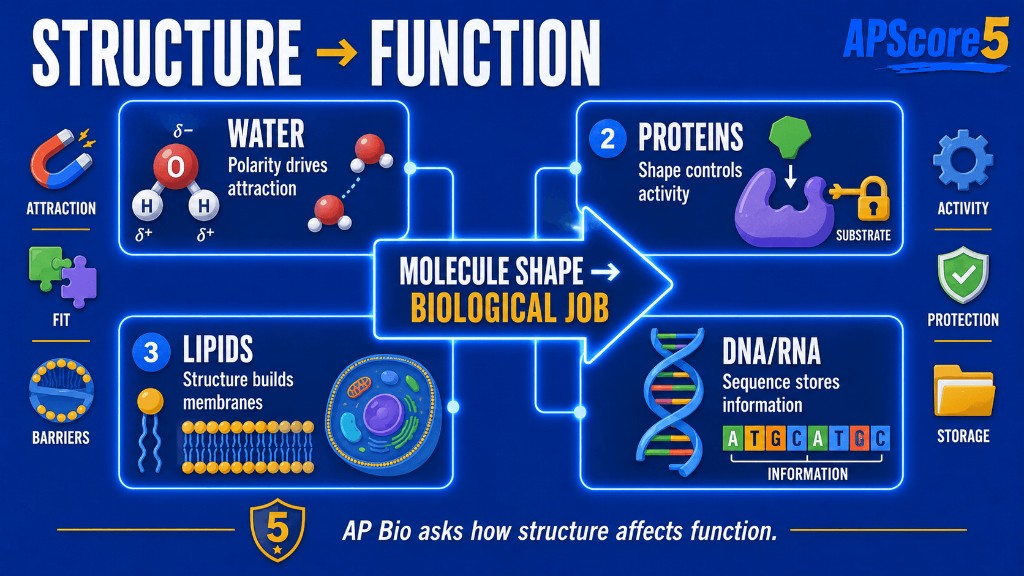
In biology, molecule shape influences what the molecule can do and how well it performs.
Large biological molecules: carbohydrates, lipids, proteins, and nucleic acids.
The two DNA strands run in opposite 5 prime to 3 prime directions.
This page tracks the Unit 1 sequence from water chemistry through nucleic acids.
Start here to see what you already know before full practice.
Hydrogen bonding gives water cohesion, adhesion, surface tension, and high specific heat, which stabilize cells and ecosystems.
Carbon can form four covalent bonds, so it creates chains and rings used in carbohydrates, lipids, proteins, and nucleic acids.
Biological molecules work because of shape. If shape changes, function can improve, weaken, or fail.
Cells build polymers through dehydration synthesis and break them by hydrolysis.
Primary to quaternary structure explains why one amino acid change can alter activity.
DNA and RNA differ in sugar, strands, and base use, but both depend on base pairing rules.
Full interactives, badge-coded review, and 16 MCQs: Properties of water microtopic →
Water is polar because oxygen pulls shared electrons more strongly than hydrogen. This partial charge difference allows hydrogen bonds between molecules. Hydrogen bonding explains cohesion in xylem columns, adhesion to vessel walls, high specific heat that buffers temperature, and surface tension that forms stable droplets.
On AP Biology questions, connect each property to a biological example. Cohesion supports upward water movement in plants, adhesion helps capillary action, and high heat capacity keeps aquatic systems from rapid temperature swings. When solutes change across a membrane, this same chemistry becomes osmosis and tonicity practice, where water movement depends on concentration gradients. That cause-and-effect explanation is what earns points.
Step 1: Cooling slows molecular movement. Step 2: Hydrogen bonds hold molecules in a more open lattice. Step 3: Molecules spread farther apart than in liquid water. Step 4: Density decreases. Since lower-density solids float on higher-density liquids, ice stays on top. Biologically, this insulates lakes, so water below remains liquid and supports life in winter.
Step 1: Transpiration at leaf surfaces pulls water upward. Step 2: Cohesion keeps water molecules linked through hydrogen bonds. Step 3: Adhesion to xylem walls helps resist gravity. Step 4: Continuous pull moves water and dissolved minerals from roots to leaves. This mechanistic chain is a frequent AP explanation target in plant transport questions.
Most biological molecules are built from CHNOPS: carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur. For a full study guide with CHNOPS tables, flashcards, and MCQs, see the elements of life AP Biology guide.
Trace elements are needed in smaller amounts but still matter for cell function. AP exam prompts may ask why a deficiency changes enzyme performance or growth rate. Link your response to molecular structure and bonding, not just memorized definitions.
Step 1: Carbon has four valence electrons. Step 2: It forms four covalent bonds with carbon, hydrogen, oxygen, nitrogen, and others. Step 3: Those bonds support chains, branches, and rings. Step 4: Diverse skeletons support diverse function, from glucose to phospholipids to DNA backbones. This explains why carbon, not silicon, dominates cell chemistry.
Step 1: ATP stores transferable energy in phosphate-group linkages. Step 2: Nucleotides use phosphate groups to build sugar-phosphate backbones. Step 3: Without phosphorus, ATP transfer and nucleic acid structure both fail. In AP items, this helps justify why phosphorus limitation can reduce growth and cellular work.
| Macromolecule | Monomer / Unit | Main Functions | Examples |
|---|---|---|---|
| Carbohydrates | Monosaccharides | Quick energy, structural support | Glucose, starch, cellulose |
| Lipids | Glycerol + fatty acids | Long-term energy, membranes, signaling | Triglycerides, phospholipids, steroids |
| Proteins | Amino acids | Enzymes, transport, structure, signaling | Amylase, hemoglobin, collagen |
| Nucleic acids | Nucleotides | Store and transfer genetic information | DNA, RNA |
Compare by structure and function. Phospholipids matter because their hydrophilic heads and hydrophobic tails explain why membranes form in AP Biology Unit 2 cell structure and function. AP Biology questions often give a molecule description and ask you to identify which class it belongs to and why.
You are told a molecule has repeating amino acids and changes shape after a pH shift. Step 1: Repeating amino acids means protein. Step 2: Shape change with pH suggests altered tertiary interactions. Step 3: Predicted function change is likely enzyme activity loss. Correct AP logic uses evidence from monomer type and structure sensitivity.
A prompt compares sprint energy to long-term energy storage. Step 1: Carbohydrates are used quickly because pathways access glucose rapidly. Step 2: Lipids store more energy per gram for long-term use. Step 3: Explain both in one answer to earn full credit when a question asks for short-term versus long-term strategy.
Proteins are chains of amino acids. The sequence (primary structure) controls local folding (secondary), full 3D shape (tertiary), and multi-chain interactions (quaternary). Shape determines active site fit and binding strength, which is why enzyme reasoning returns in AP Biology Unit 3 cellular energetics. That is why even one amino acid substitution can change transport, signaling, or enzyme rate.
When heat or pH disrupts weak interactions, proteins can denature and lose function. AP prompts often ask how environmental change affects enzyme activity curves. Explain with active-site shape and substrate compatibility.
Step 1: A single amino acid substitution changes hemoglobin primary structure. Step 2: Altered side-chain chemistry changes intermolecular interactions. Step 3: Hemoglobin molecules can aggregate under low oxygen. Step 4: Red blood cells deform, which affects transport and circulation. This chain directly models how a sequence change can alter phenotype.
A graph shows low activity at pH 2, high at pH 7, and lower at pH 10. Step 1: Ionization states of amino acid side chains shift with pH. Step 2: Active-site geometry and charge distribution change. Step 3: Substrate binding and catalysis decline away from the optimal pH. This mechanism-based explanation scores better than “enzyme was damaged.”
DNA uses deoxyribose and bases A, T, C, G. RNA uses ribose and U instead of T. DNA is typically double-stranded with antiparallel directionality (5' to 3' opposite 3' to 5'), while RNA is usually single-stranded. Base pairing rules are A-T (or A-U in RNA) and C-G.
In AP Biology Unit 1, know these differences and explain why hydrogen bonds and strand direction matter for accurate replication and information transfer. Those same nucleic acid rules carry into gene expression and regulation, especially when you compare transcription vs translation.
If one DNA strand is 5'-A C G T T A-3', the complementary strand must be 3'-T G C A A T-5'. Step 1: Match bases using A-T and C-G. Step 2: Reverse direction for antiparallel pairing. Step 3: Confirm orientation labels. AP questions often include one wrong answer that matches bases but ignores direction.
A sample contains uracil and appears mostly single stranded. Step 1: Presence of U strongly suggests RNA. Step 2: Single-stranded structure supports that call. Step 3: If ribose is listed instead of deoxyribose, that confirms RNA. State all clues in your response to show evidence-based reasoning.
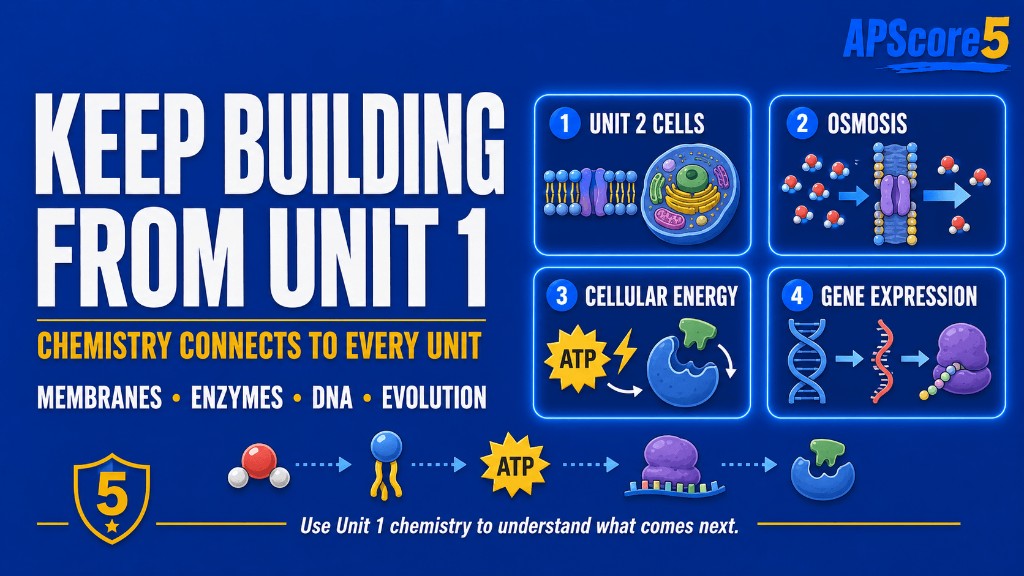
Use these next steps when Chemistry of Life starts showing up inside membranes, energy, gene expression, and evolution questions.
Once the molecule rules feel familiar, move into retrieval practice. Start with flashcards if vocabulary is shaky, or go straight to MCQs if you want exam-style reasoning and score feedback.

Every 5th card shows an ad placeholder and the next card appears after 3 seconds.
50 multiple-choice questions with score tracking. Difficulty rises through the set.
An FRQ-style prompt gives a data table comparing leaf temperature and transpiration in two habitats. Strong responses tie the properties of water to outcomes: high specific heat buffers temperature swings, and cohesion plus adhesion support continuous xylem flow. If a treatment reduces transpiration, explain how that changes water movement and thermal balance.
You may get an enzyme-rate graph with pH or temperature treatments. Explain changes by discussing active-site shape and side-chain interactions, then connect structure changes to reduced catalysis. Include one control variable and one prediction for a follow-up experiment, because AP Biology FRQs often reward experimental reasoning alongside content knowledge.
A free-response may provide a nucleotide substitution and ask for molecular consequences. Walk the chain: base change, possible codon change, amino acid effect, protein-folding effect, and potential phenotype change. If the mutation is silent, explain why amino acid sequence remains unchanged. When those molecular differences affect survival or reproduction, the reasoning continues into AP Biology Unit 7 natural selection. Clear causal links earn more points than isolated facts.
After each MCQ set, apply your knowledge using short free-response prompts modeled on the AP exam.
For every scenario, follow this exact structure:
Claim: State your answer clearly
Evidence: Cite specific data or details from the prompt
Reasoning: Explain how and why the science supports your claim
Prompt: An enzyme loses activity after a single amino acid substitution near its active site. Explain why.
Strong response
Claim: The mutation changes the enzyme's function because it alters active-site shape.
Evidence: Amino acid substitutions can change R-group interactions that stabilize tertiary structure.
Reasoning: If the active site no longer fits the substrate precisely, binding and catalysis both decline, so reaction rate drops even when substrate is present.
AP graders score more than a final answer-they score how well you use evidence, apply mechanisms, and communicate reasoning clearly.
Practicing this structure after MCQs builds the exact skills needed to earn full points on test day.
Water polarity, hydrogen bonding, and the properties of water.
CHNOPS and why carbon is central in biology.
Monomers, polymers, dehydration synthesis, hydrolysis.
Macromolecule comparison and function practice.
Protein structure and enzyme structure-function links.
DNA vs RNA, antiparallel strands, base pairing.
Mixed review: 20 flashcards + 20 practice questions.
Build cumulative accuracy by mixing Unit 1-1 concepts each day instead of reviewing one section in isolation.
Students can find AP Bio Unit 1 materials on other study platforms. This page includes 60 flashcards with explanations, 50 MCQs with answer reasoning, and FRQ-style scenarios in one study flow.
Use original AP-style practice questions with explanations instead of looking for secure test answers. APScore5 practice questions include answer reasoning so you can learn from mistakes and review weak Unit 1 topics.
The best way to review AP Biology Unit 1 is to follow the Chemistry of Life learning path, review weak topics, practice MCQs, and write short FRQ explanations using structure-function reasoning.
The set starts with straightforward recall and builds toward multi-step reasoning so you can practice the same progression seen in exam-level multiple-choice sections.
Use the topic guides on the learning path, section summaries, flashcards, and practice explanations on this page as a complete AP Bio Unit 1 study guide.
Unit 1 covers water properties, CHNOPS, monomers and polymers, dehydration synthesis and hydrolysis, and biological macromolecules — carbohydrates, lipids, proteins, and nucleic acids — with structure-function reasoning throughout.
Work through each guide sequentially — Water, Elements, Monomers, Reactions, then macromolecules, review, and practice.
Keep your momentum. Continue directly into Unit 2 so molecular chemistry connects to membranes and cell structure.