What is osmosis and tonicity in AP Biology?
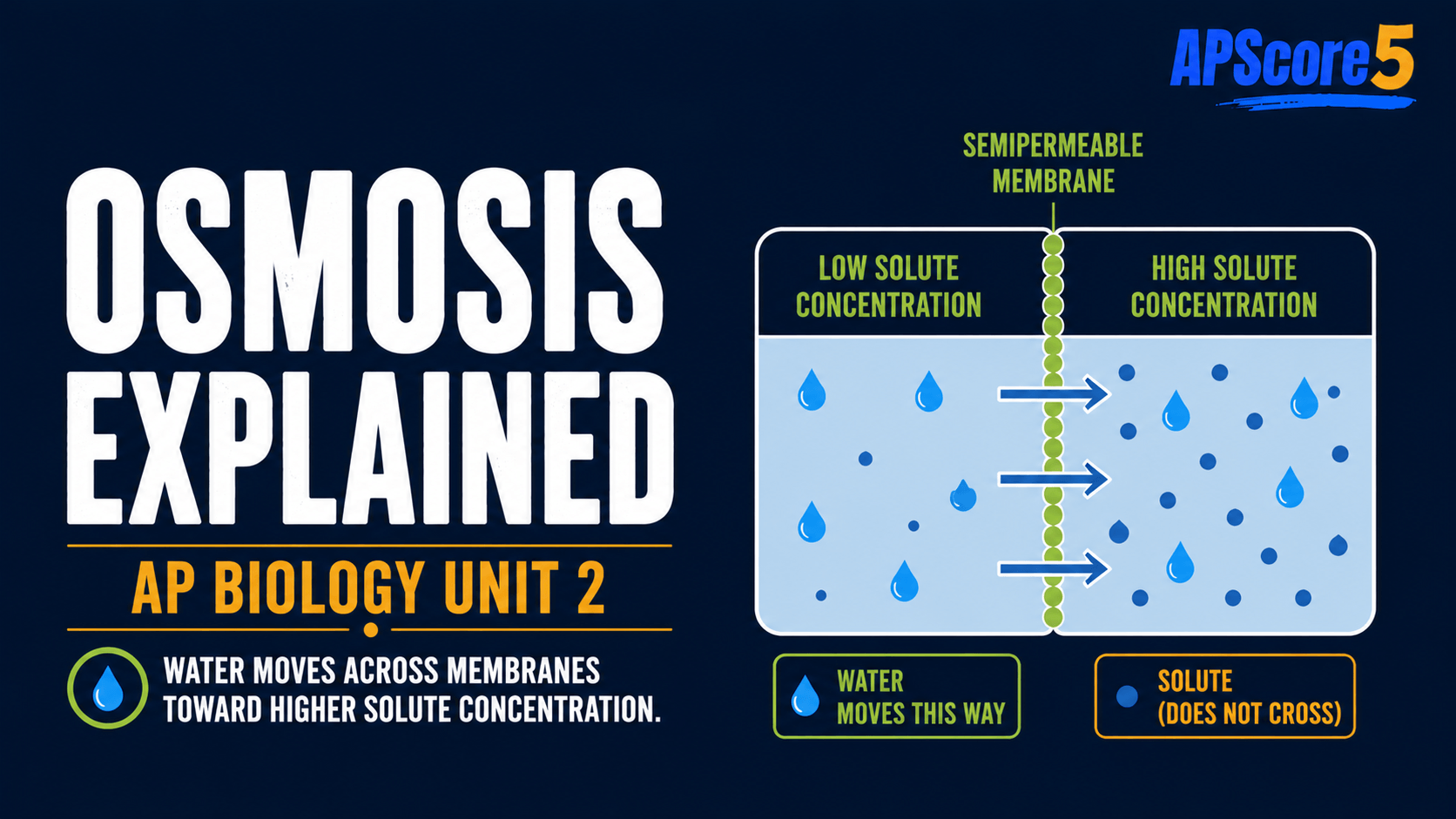
Osmosis is passive movement of water across a selectively permeable membrane from higher water concentration to lower water concentration. Tonicity compares the outside solution to the cell: hypotonic, hypertonic, and isotonic describe net water movement, and on AP Biology water flows from higher water potential Ψ to lower Ψ.
In one sentence: Osmosis is the movement of water across a selectively permeable membrane, and tonicity predicts whether a cell will gain water, lose water, or stay the same size.
What shorthand phrases matter on test day?
Key idea: Tonicity AP Biology labels describe the solution outside the cell compared with the cell—not the cell itself. Pair those labels with selective permeability and aquaporins when you explain why water crosses the membrane.
AP Must Know
- Osmosis is passive transport of water.
- Water moves across selectively permeable membranes.
- Water moves from higher water potential to lower water potential.
- Hypotonic solution: animal cells swell and may undergo cytolysis (lysis).
- Hypertonic solution: animal cells shrink (crenation).
- Isotonic solution: no net water movement.
- Plant cells become turgid in hypotonic solutions.
- Plant cells plasmolyze in hypertonic solutions.
- Water potential is calculated as Ψ = Ψs + Ψp.
Add these badges beside the Unit 2 tonicity recap before potato osmosis lab or dialysis tubing osmosis lab FRQs where mass change tracks Ψ.
How do osmosis and tonicity look at a glance on the exam?
You will switch between words (“more solute outside”) and numbers (negative Ψ). Keep the color badges in your notes:hypotonic solution → water tends to enter;isotonic solution → no net gain or loss;hypertonic solution → water tends to exit.
Table caption: Use this osmosis and tonicity AP Biology table to compare ideas you must define on FRQs.
| Concept | Microscopic definition | AP reasoning payoff |
|---|---|---|
| Osmosis | Diffusion of water across selectively permeable barriers | Shows passive transport without ATP |
| Tonicity | Relative effect of the external fluid on cell water balance | Shows you labeled the bath, not the cytoplasm |
| Water potential (Ψ) | Combines solute potential and pressure potential | Gives numeric proof for direction of water movement |
| Selective permeability | Lets water through while restricting many solutes | Explains potato osmosis lab and dialysis tubing osmosis lab setups |
Why it matters: Water moves toward the side with lower water potential (or higher effective solute concentration when the membrane blocks those solutes). Say which membrane and which solutes can cross before you call a bath hypo, iso, or hyper.
What is osmosis at the molecular scale?
Osmosis AP Biology means water moves passively—no ATP—through the lipid bilayer and through aquaporins. Random molecular motion drives net flow until gradients balance or something physical stops the cell from changing shape.
Why it matters: You must name the membrane and which solutes can cross. If a solute can diffuse through the membrane, a simple “hypotonic/hypertonic” story may fail—osmosis follows effective water availability, not just the label on the beaker.
- Osmosis is diffusion of H2O specifically.
- Aquaporins speed water movement; some water still crosses without them.
- Net flow stops when Ψ balances or when walls/turgor limit swelling.
- Saying “water moves toward higher solute” only works when that solute cannot cross.
Connect transport types on the AP Biology Unit 2 transport overview; active pumps are a different topic.
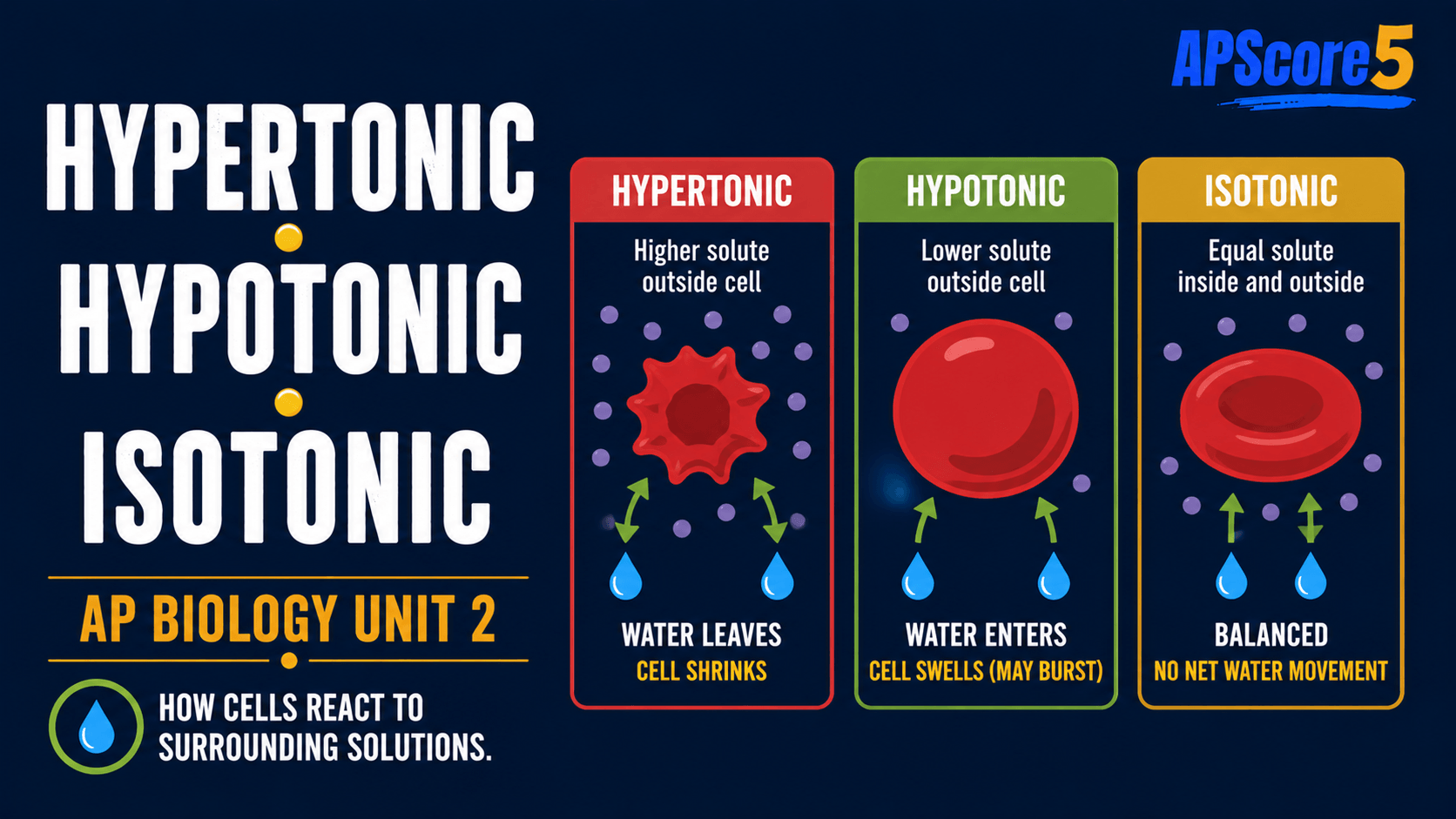
What is tonicity when solutions surround cells?
Hypotonic solution: lower effective solute outside (relative to the cell). Isotonic solution: balanced. Hypertonic solution: higher effective solute outside. Labels compare the bath to the cell under the problem’s membrane rules.
Tonicity in 3 Lines
- Hypotonic: lower solute outside → water enters → animal cell may lyse; plant cell becomes turgid.
- Isotonic: equal effective solute → no net water movement → stable volume.
- Hypertonic: higher solute outside → water exits → animal cell crenates; plant cell plasmolyzes.
Table caption: Quick reference for hypotonic hypertonic isotonic outcomes—always tie the row to the cell type in the prompt.
| Fluid label | Solute footing | Net water movement | Classic morphology |
|---|---|---|---|
| Hypotonic | Lower concentration outside | Water enters the cell | Swollen animal cells; turgid plant cells |
| Isotonic | Matched effective strength | No sustained net flow | Stable red blood cells; flaccid plant tissue |
| Hypertonic | Higher solute outside | Water leaves the cell | Crenated red blood cells; plasmolyzed plant cells |
Memory aid: hypo = “under” the outside solute tally; hyper = above it; iso = same effective balance for the comparison cell.
Why it matters: The same bottle can act hypo or hyper relative to different cells if cytoplasm tonicity differs—read the passage carefully.
Why does water move between compartments?
Water potential AP Biology (Ψ) combines every factor that changes how “free” water is: dissolved solutes (Ψs), physical pressure (Ψp), and sometimes gravity in tall columns.
Pure water at standard pressure is defined as Ψ = 0 MPa—a reference point. Adding solute makes Ψ more negative. Positive pressure (like turgor in a plant vacuole) raises Ψ.
Two beakers can look different and still have the same Ψ. Two solutions with the same molarity can behave differently if one solute crosses the membrane and another does not—predict from effective permeability.
In xylem, strong tension can make Ψp very negative; in root cells, positive turgor pushes Ψ back up. Same symbols, different story—match the scenario.
Hydrogen bonding between water molecules—your bridge back to hydrogen bonding in water properties—sets up why gradients move across membranes instead of instantly equalizing.
Leaf cells lose turgor when transpiration pulls harder than root uptake; daytime water splitting in photosynthesis and plant water balance sits upstream of many tonicity stories.
Mitochondria burn imported sugars using osmotically controlled compartments—review how cellular respiration examples across tissues tie ATP demand to ion pumping.
Test yourself #1
A red blood cell is placed in pure distilled water (a hypotonic solution). What will most likely happen?
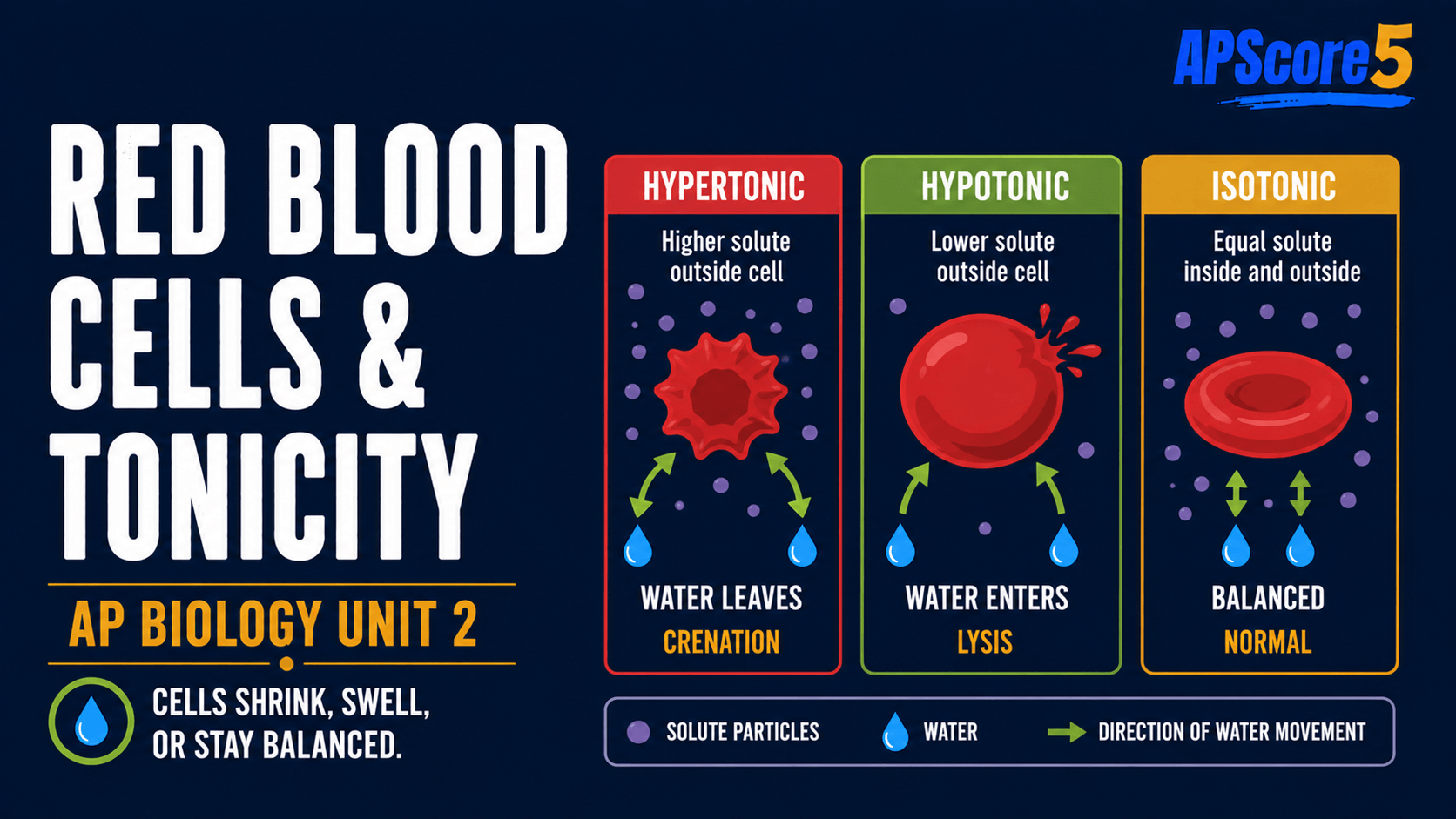
How do animal cells respond to tonicity?
Without a wall, an animal cell depends on the membrane alone. In a hypotonic solution, water enters, the cell swells, and it may burst—cytolysis (also called hemolysis in red blood cells).
In isotonic saline (~0.9% NaCl), red blood cells keep a normal disc shape. In a hypertonic solution, water leaves, and cells shrink and scallop—crenation.
Animal cell cheat sheet
- Hypotonic → water enters → swelling / lysis.
- Isotonic → no net movement → normal shape.
- Hypertonic → water leaves → crenation.
Table caption: This animal cell tonicity table shows why red blood cells lyse in hypotonic solutions and crenate in hypertonic solutions.
| Bathing fluid | RBC phenotype | Vocabulary payoff |
|---|---|---|
| Hypotonic | Swollen, rounding, may burst | Cytolysis / hemolysis |
| Isotonic (~0.9% NaCl) | Stable biconcave disc | Clinical equilibrium |
| Hypertonic | Shrunken, spiky edges | Crenation |
Exam tip: Freshwater across respiratory surfaces can drive excess water into tissues; salty environments pull water out—same Ψ logic, different organs.
Try the tonicity simulator
Select animal or plant and a bath label, then run the animation to rehearse net water movement—match every MCQ to animal cell vs plant cell outcomes.
How do plant cells respond to tonicity?
A rigid cell wall changes everything. In a hypotonic solution, water enters, the vacuole pushes outward, and the cell becomes turgid—turgor pressure holds the plant upright.
Plant cell cheat sheet
- Hypotonic → water enters → turgid.
- Isotonic → no net gain → flaccid.
- Hypertonic → water leaves → plasmolysis.
Table caption: This plant cell tonicity table shows why plant cells become turgid in hypotonic solutions and plasmolyze in hypertonic solutions.
| External tonic label | Typical plant cell look | Formal vocabulary |
|---|---|---|
| Hypotonic | Membrane pressed against the wall | Turgid; positive Ψp from turgor |
| Isotonic (balanced) | Less firm leaf tissue | Flaccid |
| Hypertonic | Membrane pulls away from the wall | Plasmolysis; wilting risk |
Why it matters: Night watering can restore turgor before midday transpiration—still the same osmosis rules.
What is water potential on AP exams?
Water potential equation: Ψ = Ψs + Ψp. Use MPa in most classroom problems.
- Ψ = total water potential.
- Ψs = solute potential; zero or negative—more solute makes Ψ more negative.
- Ψp = pressure potential; positive, zero, or negative—turgor pressure makes Ψp more positive in plant cells.
Rule: Water moves from higher Ψ to lower Ψ. Pure water at standard pressure has Ψ = 0 MPa.
This water potential equation is the fastest way to defend direction on FRQs—always show your sum before you narrate swelling or plasmolysis.
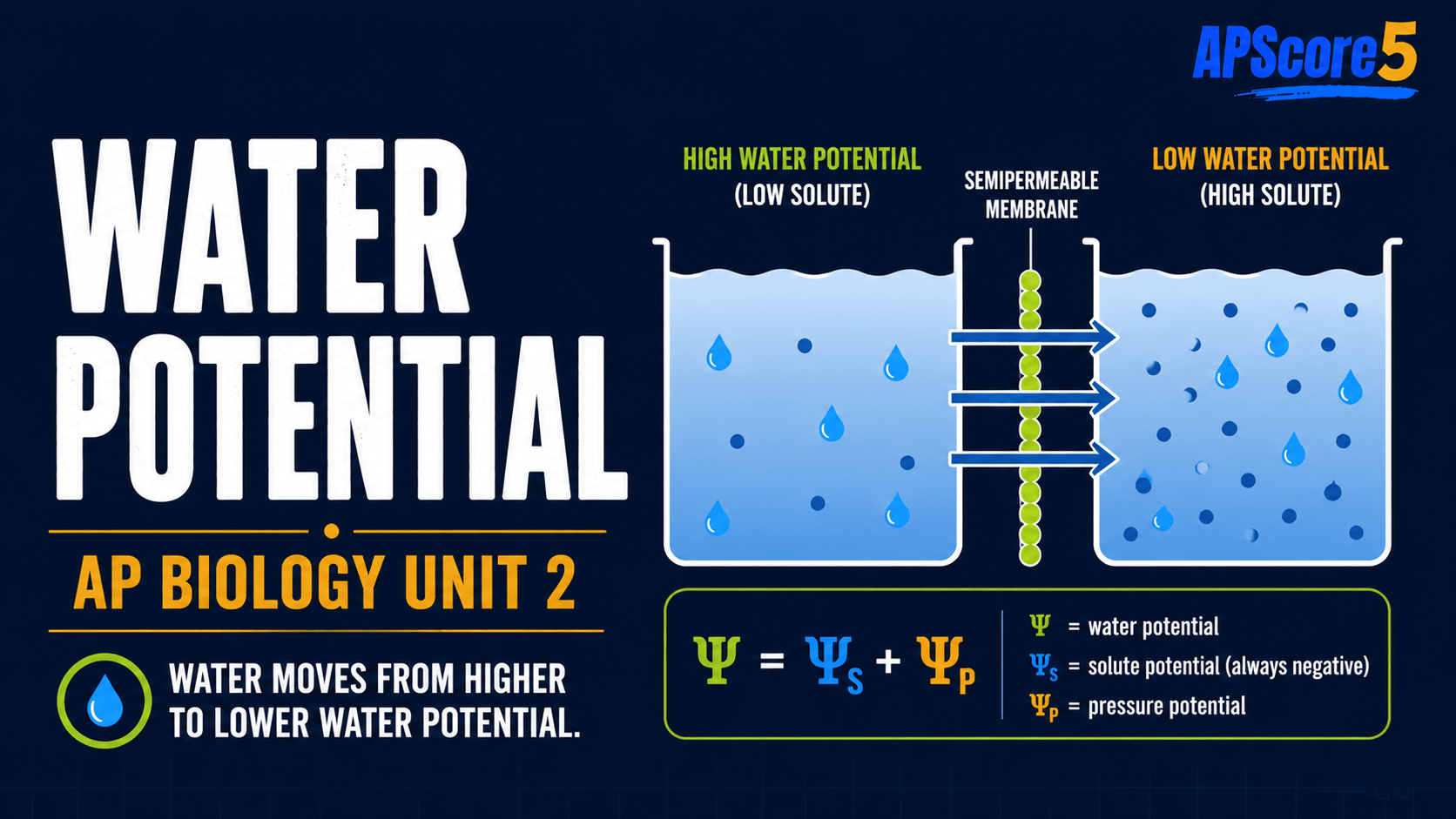
Table caption: This water potential AP Biology table explains solute potential, pressure potential, and total Ψ.
| Component | Meaning | Sign practice |
|---|---|---|
| Ψs (solute potential) | Solute lowers free water; ties to concentration | ≤ 0 when solutes are present |
| Ψp (pressure potential) | Physical push or pull on water | Often positive for turgor; very negative in xylem tension |
How do you crunch a stacked water potential FRQ?
- Copy Ψs and Ψp with their signs.
- Add to get total Ψ for the cell.
- Compare compartments: water moves from higher Ψ to lower Ψ.
Sample prompt slice: Plant cortex has Ψs = −1.5 MPa and Ψp = +0.3 MPa, so Ψ = −1.2 MPa. A bath at Ψ = −0.8 MPa is higher than −1.2 MPa, so water still enters even though both numbers are negative.
If the bath is Ψ = −2.6 MPa, water leaves the cell—useful for explaining plant cells undergoing plasmolysis in FRQ microscopy prompts.
Water potential sandbox calculator
Type MPa placeholders exactly as workbook prompts dictate. Optional bathing Ψ activates comparative narration.
Test yourself #2
A plant cell has Ψ = −1.2 MPa. It is placed in a solution with Ψ = −0.5 MPa. What will happen?
How does osmosis compare to diffusion?
Osmosis vs diffusion: Diffusion is movement of any molecule down its concentration gradient. Osmosis is the diffusion of water across a selectively permeable membrane—same passive idea, stricter setup.
Table caption: This osmosis vs diffusion table separates water movement from general molecule movement.
| Contrast axis | Diffusion example | Osmosis example |
|---|---|---|
| Molecule | O2, CO2, ions (depends on pathway) | Water |
| Barrier | May be open solution | Needs a selectively permeable membrane |
| Driver language | Concentration gradient | Ψ gradient / effective water availability |
| Energy | Passive | Passive |
| Lab tie-in | Perfume spreading in air | Dialysis tubing osmosis lab; potato osmosis lab |
Key idea: All osmosis is diffusion, but not all diffusion is osmosis.
What everyday scenarios rehearse hypo, iso, hyper logic?
Use quick stories to check your reasoning before you write a long FRQ paragraph.
Table caption: Real observations mapped to osmosis AP Biology vocabulary—name whether the outside fluid acts like a hypotonic solution, isotonic solution, or hypertonic solution for the cells in the story.
| Observation | Interpretation shorthand |
|---|---|
| Wilting pothos midday | Soil water potential drops; root cells lose turgor (outside acts hypertonic) |
| Lettuce perks up in clean water | Hypotonic rinse pushes water in; Ψp rises |
| Raisins plump in a water bowl | Water is hypotonic to the dried fruit cells |
| Salty jerky curing | Hypertonic salt pulls water from microbes |
| IV fluid matched to plasma | Isotonic bags protect red blood cells |
| Freshwater fish produce lots of dilute urine | Environment is hypotonic; excess water enters |
| Many marine fish drink seawater and excrete salt | Ocean reads hypertonic; salt regulation adds active transport |
Why it matters: Irrigation timing is about keeping soil Ψ in a range where crop roots stay turgid—same math as a potato osmosis lab, just at field scale.
Test yourself #3
If you place a peeled potato slice in concentrated saltwater for 30 minutes, what will happen to the potato?
How do osmosis and tonicity appear on AP Biology prompts?
Expect MCQs with micrographs—plant epidermis showing plasmolysis vs red blood cells showing lysis or crenation—and quick tonicity labels tied to saline or soil contexts.
FRQs often combine Ψ arithmetic with explanation: show addition for Ψ = Ψs + Ψp, compare baths, then describe turgor loss or cell bursting.
Why it matters: Earn reasoning points by citing both numbers and cell-type outcomes (wilting leaves vs lysed blood cells).
AP Exam Answer Template
Water moves from higher water potential to lower water potential. Because the outside solution is [hypotonic / isotonic / hypertonic] relative to the cell, water will move [into / out of / neither direction overall], causing the cell to [swell / shrink / stay stable / become turgid / plasmolyze].
What mistakes cost students points most often?
- Saying “water moves to high water concentration” while your math shows movement toward lower Ψ—keep wording and symbols aligned.
- Forgetting that some solutes cross membranes—tonicity fails if urea can diffuse while sucrose cannot.
- Treating pure water as negative Ψ—reference pure water at Ψ = 0 MPa when the problem uses that convention.
- Mixing up plasmolysis (plant) with crenation (animal red blood cells).
- Claiming osmosis uses ATP—it is passive transport.
- Calling two baths “hypertonic” from labels alone when permeability changes who actually has higher effective solute.
- Describing turgid plant cells as “burst”—walls prevent bursting; turgor is normal.
Flip through 22 osmosis and tonicity cards
AP Biology styled multiple-choice (16 prompts)
Osmotic onion peel lab storyline
An investigator swaps onion peels among distilled water vs sucrose reservoirs. Beginning Ψs = −1.5 MPa and Ψp = +0.3 MPa. Bathing solutions expose Ψ = 0 MPa, −1.0 MPa, and −2.5 MPa respectively.
(A) Define Ψ and explain both Ψs and Ψp.
(B) Compute initial cortical Ψ with shown arithmetic.
(C) Predict water direction for each bath using Ψ comparisons.
(D) Relate long exposure in the −2.5 MPa bath to wilting in the field.
Osmosis and tonicity FAQs
What is osmosis?
Osmosis is diffusion of water across a selectively permeable membrane from higher water concentration to lower water concentration. It is passive transport (no ATP). Water crosses the lipid bilayer and moves faster through aquaporins.
What is tonicity?
Tonicity compares the outside solution to the cell and predicts net water movement based on effective solute concentrations across the membrane.
What is the difference between hypotonic, isotonic, and hypertonic solutions?
Hypotonic solutions have lower effective solute outside (relative to the cell), so water tends to enter. Isotonic solutions match effective solute, so there is no net water movement. Hypertonic solutions have higher effective solute outside, so water tends to leave.
What happens to animal cells in a hypotonic solution?
Water enters the cell. Without a cell wall, the cell swells and may undergo cytolysis (bursting); red blood cells undergo hemolysis.
What happens to animal cells in a hypertonic solution?
Water leaves the cell, so it shrinks. Red blood cells often look scalloped or spiky, called crenation.
What happens to plant cells in a hypotonic solution?
Water enters, turgor pressure rises against the cell wall, and the plant cell becomes turgid; this is usually healthy for plants.
What happens to plant cells in a hypertonic solution?
Water leaves the cell, the membrane pulls away from the wall, and the cell undergoes plasmolysis.
What is plasmolysis?
Plasmolysis is shrinkage of the plant cell membrane away from the cell wall after water loss in a hypertonic solution.
What is turgor pressure?
Turgor pressure is outward pressure of water against the plant cell wall from water entering the central vacuole; it keeps tissues firm and supports stomatal opening.
What is water potential?
Water potential (Ψ) predicts where water moves. It combines solute potential and pressure potential in Ψ = Ψs + Ψp. Water moves from higher Ψ to lower Ψ.
How do you calculate water potential?
Use Ψ = Ψs + Ψp with consistent units (often MPa). Add solute potential and pressure potential for each compartment, then compare totals.
What is solute potential?
Solute potential (Ψs) reflects dissolved solutes; it is zero or negative, and more solute usually makes Ψ more negative.
What is pressure potential?
Pressure potential (Ψp) reflects physical pressure on water; it can be positive (turgor), zero, or negative (tension), depending on the scenario.
What is the difference between osmosis and diffusion?
Diffusion is movement of any molecule down its concentration gradient. Osmosis is diffusion of water across a selectively permeable membrane. Osmosis vs diffusion is about water plus the membrane requirement.
Is osmosis active or passive transport?
Osmosis is passive transport; it does not require ATP because water moves down its water potential gradient.
What does a potato osmosis lab show?
Changing outside sucrose concentration changes bath water potential, so potato cells gain or lose water and core mass changes track net osmosis; dialysis tubing osmosis labs use the same selective-permeability logic.