What are water properties in AP Biology?
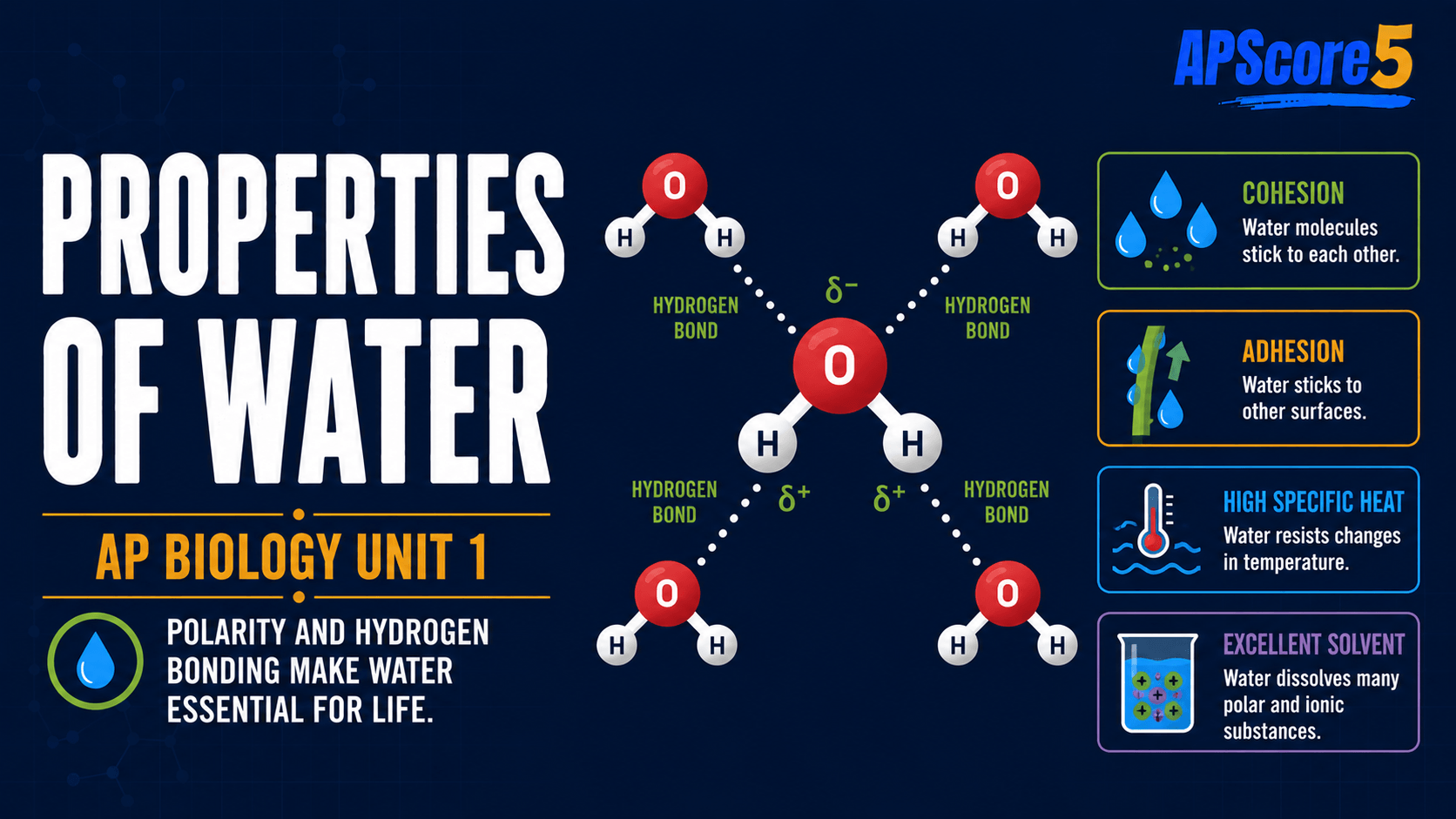
The six major properties are cohesion, adhesion, high specific heat, high heat of vaporization, lower density as a solid (ice floats), and universal solvent behavior—your core properties of water AP Biology list. Water polarity and hydrogen bonding in water explain them, enabling transport, temperature stability, evaporative cooling, insulated aquatic habitats, and dissolving ions and polar molecules.
In one sentence: Water’s polarity and hydrogen bonding give it life-supporting properties such as cohesion, adhesion, high specific heat, evaporative cooling, ice floating, and solvent behavior.
How should you introduce water on exam day?
Key idea: Start with polarity, then name hydrogen bonding in water, then connect each macroscopic trait to a living-system example—water properties examples graders love include xylem, sweating, and pond ice.
AP Must Know
- Water is polar because oxygen is more electronegative than hydrogen.
- Water has a bent shape, so partial charges do not cancel.
- Hydrogen bonds form between different water molecules.
- Cohesion means water sticks to water.
- Adhesion means water sticks to other polar surfaces.
- High specific heat helps stabilize temperature.
- High heat of vaporization allows evaporative cooling.
- Ice floats because solid water is less dense than liquid water.
- Water dissolves polar and ionic substances.
Properties of water at a glance
Use this grid as your water properties AP Bio checklist—each row links a term to real biology.
6 Properties at a Glance
- Cohesion: water sticks to water → surface tension and xylem columns.
- Adhesion: water sticks to surfaces → capillary action.
- High specific heat: resists temperature change → stable bodies and climates.
- High heat of vaporization: evaporation removes heat → sweating and transpiration.
- Ice floats: solid less dense → aquatic habitats stay insulated.
- Universal solvent: dissolves polar and ionic substances → transport in cells and blood.
Table caption: Use this properties of water AP Biology table to connect each property to hydrogen bonding and a biological example.
| Property | Meaning | Life payoff |
|---|---|---|
| Cohesion | Water attracts itself | Surface tension; continuous xylem columns |
| Adhesion | Water clings to polar surfaces | Capillary rise; wall wetting |
| High specific heat | Temperature shifts slowly | Stable bodies and coastal climates |
| High heat of vaporization | Vaporizing steals massive energy | Sweating and transpiration cooling |
| Less dense ice | Solid floats on liquid | Insulating lid on lakes |
| Universal solvent | Dissolves polar and ionic species | Blood chemistry and cytosolic reactions |
Why is water central to living systems?
Cells are roughly 60–70% water. Metabolism, ion transport, and enzyme chemistry all assume an aqueous medium—lose water and proteins unfold, membranes fail, and plants lose turgor.
Water also joins reactions such as photolysis in photosynthesis and hydrolysis patterns tied to cellular respiration. For movement into cells, pair this page with osmosis and tonicity.
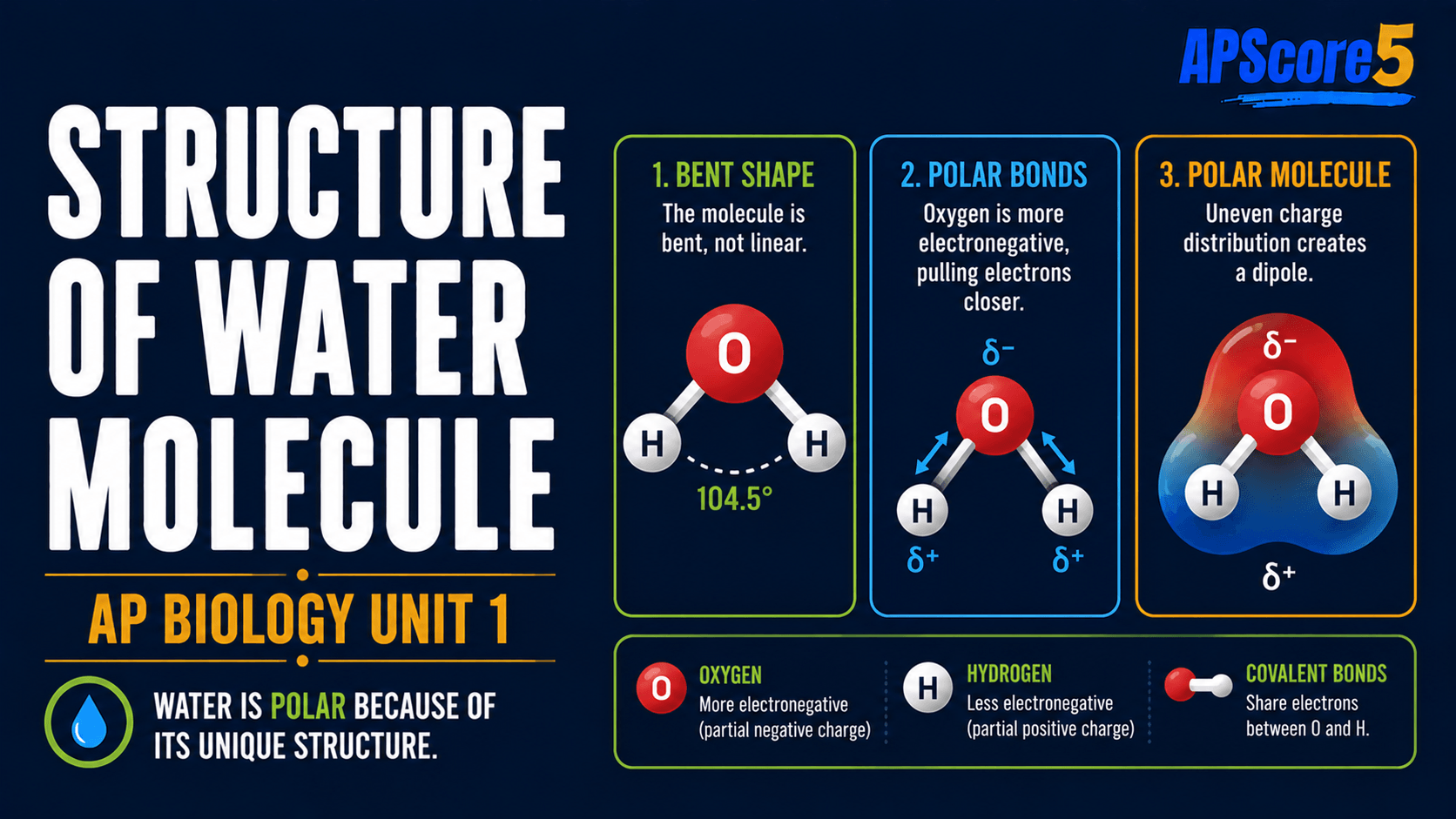
How is a single water molecule organized?
Polar covalent bonds in water: Each O–H bond shares electrons unevenly—oxygen attracts shared electrons more strongly than hydrogen.
- Oxygen carries a partial negative charge (δ−); hydrogens carry partial positive charges (δ+).
- Water is bent (about 104.5°), not linear, so charges do not cancel—central to water polarity AP Biology reasoning.
- Lone pairs on oxygen can accept hydrogen bonding in water from neighbors.
Caption: This water polarity checklist pairs bent shape with uneven charge sharing.
Compare with linear CO2: symmetry lets dipoles cancel, so CO2 is nonpolar despite polar bonds—shape decides water polarity outcomes on MCQs.
Explore partial charges on one molecule
Pointer or keyboard focus highlights δ regions without leaving the figure.
Hover or tap oxygen, hydrogens, bonds, or angle labels.
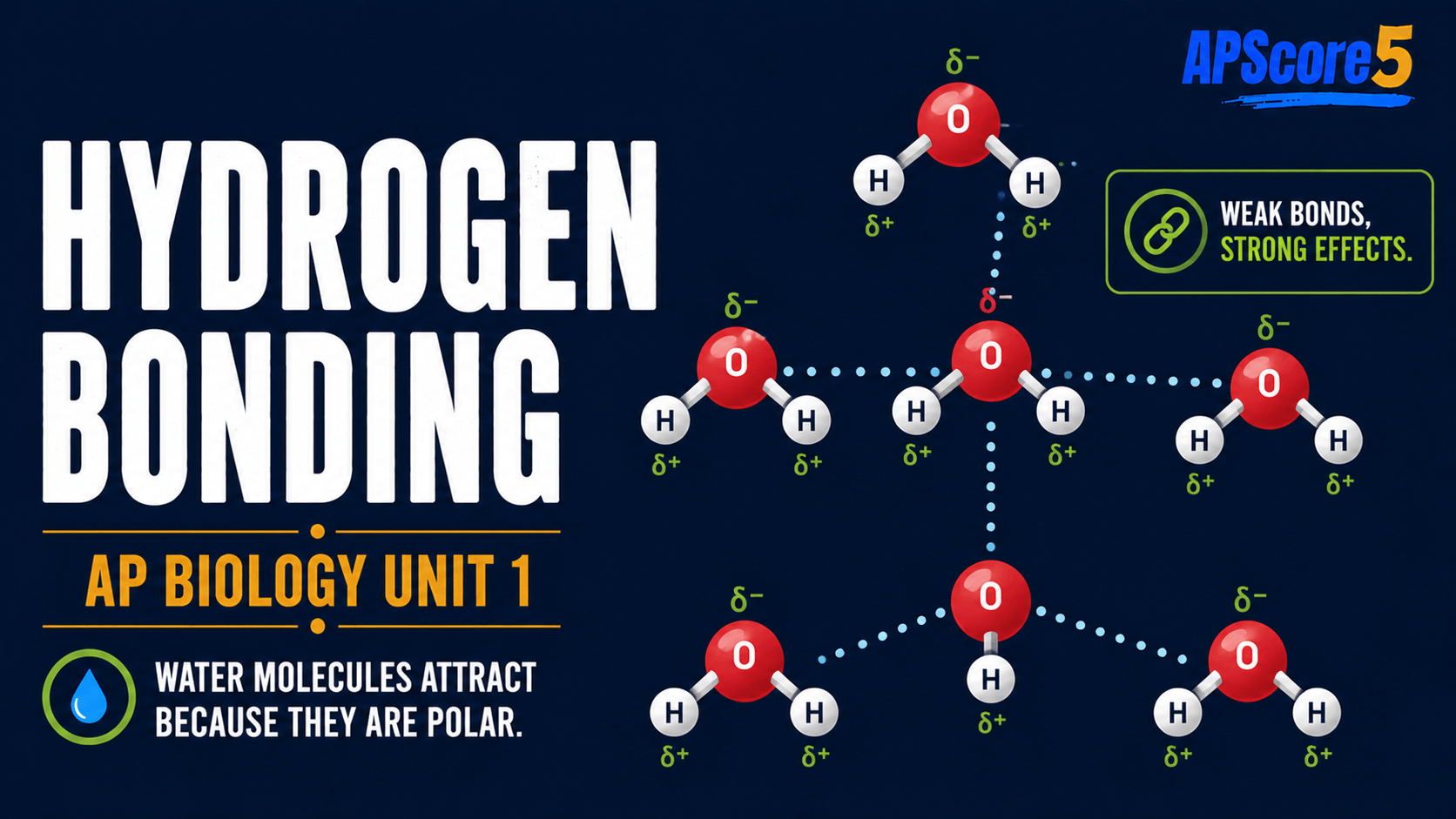
How do hydrogen bonds arise between waters?
δ+ hydrogens on one molecule align toward δ− oxygen on another. One hydrogen bond is weak, but many hydrogen bonds act together—that scale effect drives cohesion, high specific heat, and vaporization costs.
Hydrogen bonds vs covalent bonds: Polar covalent bonds hold O and H inside a single molecule; hydrogen bonds form between separate water molecules (and between water and other polar groups).
Table caption: This hydrogen bonds vs covalent bonds comparison helps avoid a common AP Biology mistake.
| Bond type | Where | Typical strength idea |
|---|---|---|
| Polar covalent (O–H) | Inside one H2O molecule | Strong intramolecular sharing |
| Hydrogen bond | Between separate water molecules | Weak alone; powerful in huge numbers |
Those same hydrogen-bond ideas extend to DNA and proteins—see Unit 1 nucleic acids and the structure-function anchor.
Hydrogen bond network flicker
Bars brighten and dim to show hydrogen bonds forming and breaking constantly in liquid water.
Dashed ties pulse to suggest bonds forming and breaking every picosecond in liquid water.
What is cohesion?
Cohesion means water sticks to water through hydrogen bonding in water networks.
- Water sticks to itself.
- Creates surface tension at the air–water boundary.
- Helps maintain continuous water columns in xylem (with tension from transpiration).
Surface tension: Top-layer molecules have no neighbors above them, so they pull sideways and downward—enough to support some insects on the surface tension “skin.”
Cohesion-tension: Transpiration pulls on a continuous water thread; cohesion keeps that thread from snapping apart.
What is adhesion?
Adhesion is water sticking to other polar surfaces—cellulose, glass, soil particles—not to another water molecule.
- Water sticks to other polar surfaces.
- Helps capillary action pull water into narrow spaces.
- Helps water cling to xylem walls beside air-filled pits.
A meniscus curves where adhesion at the wall meets cohesive inward pull.

How do cohesion and adhesion differ on FRQs?
Graders check cohesion vs adhesion partners: H₂O–H₂O versus H₂O–surface.
Table caption: This cohesion vs adhesion table separates water-to-water attraction from water-to-surface attraction.
| Feature | Cohesion | Adhesion |
|---|---|---|
| Partners | Water–water | Water–solid or polar surface |
| Exam buzzwords | Surface tension, continuous column | Meniscus, wall wetting |
| Plant context | Maintains sap chain under tension | Wets xylem walls; supports capillary rise |
Mnemonic: cohesion ≈ “co-water”; adhesion attaches elsewhere.
Test yourself #1
A water strider walks on the surface of a pond without sinking. Which property of water makes this possible?
Why does water resist rapid heating?
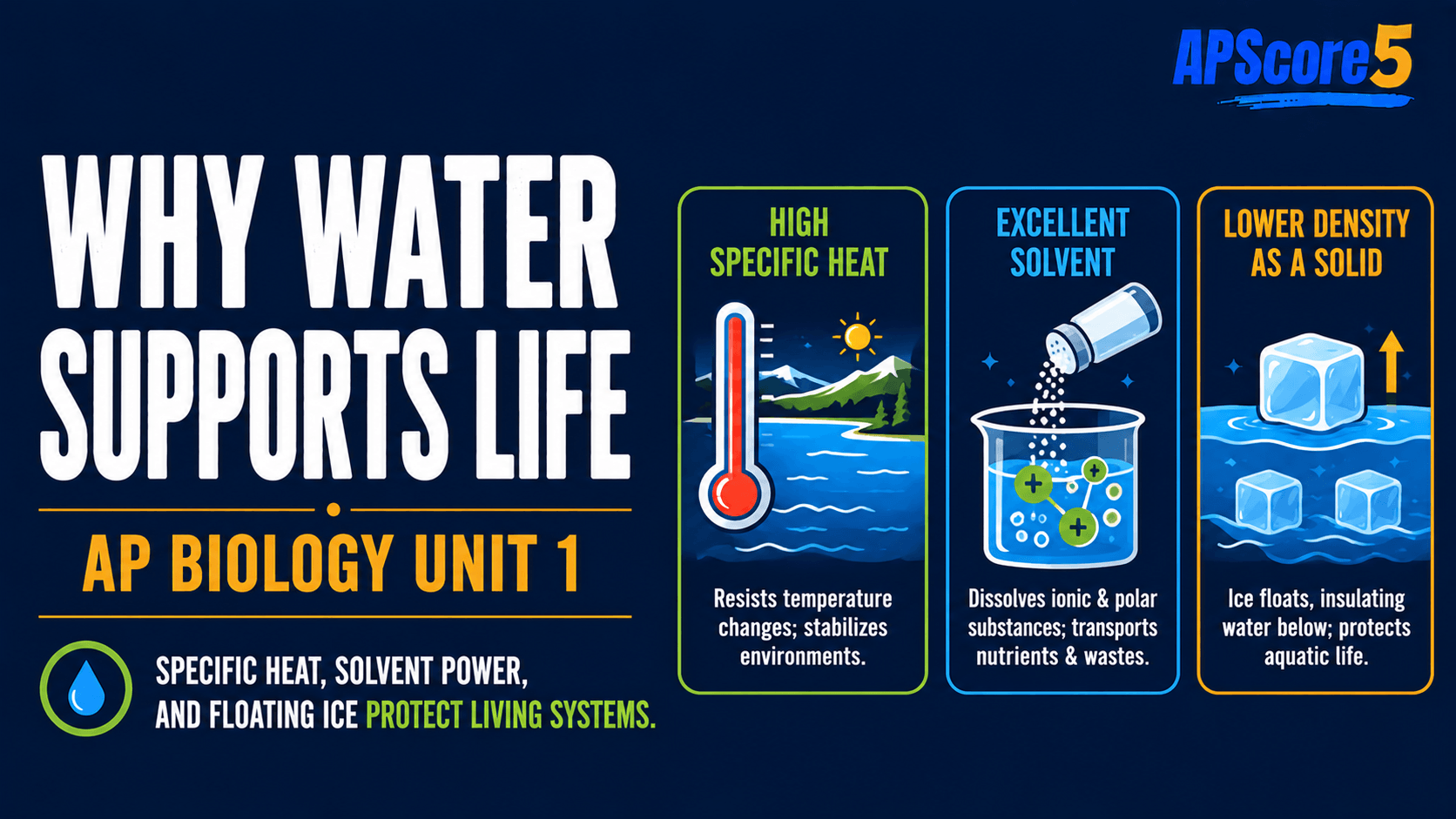
High specific heat means water absorbs lots of energy before its temperature rises much: added heat first breaks hydrogen bonding in water; only leftover energy speeds up molecular motion (temperature).
- Water resists temperature change.
- Stabilizes body temperature and large aquatic habitats (high specific heat of water).
Oceans and blood plasma moderate swings because high specific heat of water slows heating and cooling.
Why does evaporation steal so much heat?
High heat of vaporization means turning liquid water into vapor costs a large energy payment to break hydrogen bonds.
- Evaporation removes heat from the surface that loses the molecules.
- Powers evaporative cooling in sweating and transpiration.
Evaporative cooling: When sweat or leaf water evaporates, energy leaves skin or leaf tissue—linked directly to heat of vaporization water demands.
Why does ice float?
Ice density answers why does ice float: freezing locks molecules into an open hydrogen-bond lattice with more space than liquid packing.
- Ice is less dense than liquid water (~9% for freshwater ice).
- Floating ice insulates liquid below—fish survive winter under the lid.
If ice sank, seasonal freezing could kill bottom communities—compare with most solids that sink when they crystallize.
Why call water the universal solvent?
Universal solvent describes water as a universal solvent for polar and ionic substances—δ− oxygen and δ+ hydrogen orient around ions and polar groups.
- Water dissolves polar and ionic substances.
- Does not dissolve nonpolar oils well—foundation for hydrophilic vs hydrophobic logic.
Preview membranes via hydrophobic clustering in Unit 1 macromolecules.
Test yourself #2
During a hot day, you sweat heavily and feel cooler. Which water property is responsible for this cooling effect?
How the properties of water enable life
Water properties examples for FRQs—pair each row with hydrogen bonding and an organism-scale payoff.
Table caption: Link each trait to how cells or ecosystems actually use properties of water AP Biology Unit 1 ideas.
| Trait | Biological example |
|---|---|
| Cohesion + tension | Tall-tree transpiration pulls continuous xylem water |
| Adhesion + cohesion | Soil water entering roots; water climbing narrow tubes |
| Specific heat | Stable body temperature; oceans moderate climate |
| Vaporization | Sweating and transpiration export heat |
| Floating ice | Liquid habitat survives under winter ice |
| Solvent behavior | Blood and cytoplasm dissolve and transport ions |
How the properties of water appear on the AP Biology exam
MCQs often test vocabulary fit—surface tension vs capillary action vs evaporative cooling. Read the stem for the scenario (pond surface, narrow tube, sweating).
FRQs usually want three layers: name the property, explain hydrogen-bond logic, give a biological consequence.
Multiple-choice traps: Ice floats → open lattice in ice (not “water shrinks when cold”); water striders → cohesion/surface tension; stable pond temps → high specific heat; cooling sweat → heat of vaporization water, not specific heat alone.
Compare nonpolar benchmarks: methane or oils lack water-like cohesion networks—say polarity is missing.
AP Exam Answer Template
Water is polar because oxygen has a partial negative charge and hydrogens have partial positive charges. This polarity allows hydrogen bonds to form between water molecules. Those hydrogen bonds explain [property], which helps living systems by [biological example].
Common AP Bio mistakes about the properties of water
These slips cost more points than forgetting a vocabulary word.
- Each hydrogen bond is weak compared with O–H polar covalent bonds inside a molecule.
- Polar covalent bonds sit inside one molecule; hydrogen bonds link separate molecules—keep labels straight (hydrogen bonds vs covalent bonds).
- Water is polar (partial charges), not an ionic crystal.
- Cohesion is water-to-water; adhesion is water-to-surface.
- High specific heat slows temperature change; heat of vaporization sets the cost to evaporate—different MCQ answers.
- Ice forms a stable hydrogen-bond lattice with extra spacing; liquid water constantly rearranges bonds.
- End with biology—ecosystem or physiology payoff.
Match scenarios to property badges
Each vignette asks which property label fits—tap fast to build automatic recall before exam-day fatigue hits.
Test yourself #3
Which combination best explains stable pond temperatures during sunny afternoons?
Properties of water flashcards (22)
Flip until you can narrate each definition with real water properties examples—speed matters less than precision on test day.
AP Biology practice MCQs (16)
AP Biology water properties practice questions—sixteen stems with typical distractors; read each explanation even when you guess correctly.
Lake ice insulating aquatic communities
A researcher observes northern ponds freezing from the top while fish remain active deeper down.
(A) Identify and define the property explaining floating ice and justify it molecularly.
(B) Explain how floating ice supports winter ecosystems.
(C) Name two additional properties of water stabilizing aquatic habitats with molecular + biological reasoning.
(D) Predict consequences if ice were denser than liquid water.
Properties of water FAQs
What are the properties of water in AP Biology?
The six major properties are cohesion, adhesion, high specific heat, high heat of vaporization, lower density as a solid (ice floats), and universal solvent behavior. They emerge from water polarity and hydrogen bonding in water.
Why is water polar?
Oxygen attracts shared electrons more strongly than hydrogen in each O–H bond, giving oxygen a partial negative charge and hydrogens partial positive charges. The bent molecular shape prevents dipoles from canceling, so water stays polar.
How do hydrogen bonds affect the properties of water?
Hydrogen bonds form between separate water molecules; many weak bonds acting together produce cohesion, surface tension, high specific heat, high heat of vaporization, floating ice, and dissolution of polar and ionic substances.
What is cohesion?
Cohesion is water sticking to water—attraction between water molecules—important for surface tension and continuous water columns in xylem.
What is adhesion?
Adhesion is water sticking to other polar surfaces—attraction between water and solids—important for capillary action and wetting xylem walls.
What is the difference between cohesion and adhesion?
Cohesion is water-to-water attraction; adhesion is water-to-surface attraction. Cohesion vs adhesion comes down to whether the partner is another water molecule or a different polar surface.
What is surface tension?
Surface tension is the cohesive tightening of the top layer of water because surface molecules lack neighbors above them; it supports small organisms like water striders.
What is capillary action?
Capillary action is water rising in narrow tubes when adhesion pulls water along walls and cohesion pulls the bulk fluid behind—used in soil pores and xylem.
Why does water have a high specific heat?
Added heat first breaks hydrogen bonds between molecules; only leftover energy raises kinetic motion (temperature). High specific heat of water slows temperature swings in oceans and organisms.
What is evaporative cooling?
Evaporative cooling happens when liquid water becomes vapor: molecules absorb heat to overcome hydrogen bonds, cooling the surface—seen in sweating and transpiration and tied to high heat of vaporization water requires.
Why does ice float on water?
Frozen water forms an open hydrogen-bond lattice with more space between molecules than liquid water, so ice is less dense and floats—insulating liquid habitats below.
Why is water called the universal solvent?
Water as a universal solvent means polar water surrounds ions and hydrogen-bonds polar molecules; nonpolar oils stay excluded. Hydration shells help blood and cytoplasm transport nutrients.
What is the difference between hydrophilic and hydrophobic?
Hydrophilic substances are polar or ionic and dissolve well in water; hydrophobic substances are nonpolar and do not—like dissolves like.
What is the difference between hydrogen bonds and covalent bonds in water?
Polar covalent bonds join oxygen and hydrogen inside one molecule; hydrogen bonds form between separate water molecules and are much weaker than covalent bonds.
Why are water properties important for life?
They stabilize temperature, dissolve and transport ions and polar metabolites, move water through plants, protect aquatic life under floating ice, and keep cellular chemistry aqueous—core to metabolism and ecosystems.