What is cellular respiration in AP Biology?
Cellular respiration is the process cells use to transfer energy from glucose into ATP. In aerobic respiration, glucose is oxidized through glycolysis, pyruvate oxidation, the Krebs cycle, and the electron transport chain. Oxygen acts as the final electron acceptor, allowing cells to make about 30–32 ATP per glucose.
Cellular respiration equation: C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + ATP
In one sentence: Cellular respiration breaks down glucose to make ATP, using glycolysis, pyruvate oxidation, the Krebs cycle, and the electron transport chain.
Plain-language snapshot
- Glycolysis happens in the cytoplasm.
- Pyruvate oxidation and the Krebs cycle happen in the mitochondrial matrix.
- The electron transport chain happens on the inner mitochondrial membrane.
- Oxygen is the final electron acceptor.
- Most ATP comes from oxidative phosphorylation.
- Fermentation regenerates NAD+ when oxygen is unavailable.
Cellular respiration overview at a glance
Use this cellular respiration AP Biology table to compare each stage by location, inputs, outputs, and ATP yield cellular respiration contributions.
About 30–32 ATP per glucose molecule under aerobic conditions
| Stage | Where it happens | Net output (per glucose) |
|---|---|---|
| 1. Glycolysis | Cytoplasm | 2 ATP, 2 NADH, 2 pyruvate |
| 2. Pyruvate oxidation | Mitochondrial matrix | 2 NADH, 2 CO2, 2 acetyl-CoA |
| 3. Krebs cycle | Mitochondrial matrix | 2 ATP, 6 NADH, 2 FADH2, 4 CO2 |
| 4. Electron transport chain | Inner mitochondrial membrane | ~26–28 ATP, 6 H2O |
- Glycolysis splits glucose in the cytosol and nets a little ATP plus NADH.
- Pyruvate oxidation feeds acetyl groups into the mitochondria while releasing CO2.
- Krebs cycling strips remaining carbons as CO2 and loads carriers.
- Oxidative phosphorylation lets electrons from those carriers drive chemiosmosis.
The stages happen in order, and each one feeds the next. Glycolysis produces pyruvate that gets oxidized into acetyl-CoA, which feeds the Krebs cycle, which produces NADH and FADH2 that fuel the electron transport chain.
Why do cells need cellular respiration?
Cells need ATP constantly. Muscle contraction, nerve signaling, protein synthesis, active transport, and macromolecule assembly all hydrolyze ATP to ADP and phosphate faster than any warehouse could store fresh ATP.
The problem is practical storage: the total ATP on hand in tissues would power movement for only seconds if replenishment stopped. Cells continuously phosphorylate ADP, and cellular respiration is the dominant refill pathway when oxygen is available.
Glucose remains a preferred fuel because it is energy-rich, water-soluble, and feeds glycolysis without elaborate preprocessing in many cell types. Roughly 40% of its chemical potential can be captured as ATP aerobically; the remainder exits as heat that helps maintain body temperature.
This connects directly to Unit 2: Cell Structure and Function: mitochondria position the Krebs enzymes in the matrix and pack electron transport plus ATP synthase into cristae so huge membrane surface area supports rapid chemiosmosis.
Contrast every oxidation story here with carbon fixation logic on the photosynthesis versus cellular respiration comparison so complementary pathways stay paired on FRQs.
Water availability changes how roots supply sugars for respiration—tie membrane math from osmosis and water potential practice to whole-plant fuel delivery.
What are the four stages of cellular respiration?
Key idea: This cellular respiration overview pairs each stage of cellular respiration with where it runs—anchor every FRQ to location before you narrate chemistry.
Think location first—two compartments dominate AP sketches: cytosol versus mitochondrial matrix versus inner membrane.
- Glycolysis: glucose → 2 pyruvate, net 2 ATP, 2 NADH
- Pyruvate oxidation: pyruvate → acetyl-CoA, CO2, NADH
- Krebs cycle: acetyl-CoA → CO2, NADH, FADH2, small ATP
- ETC + chemiosmosis: NADH/FADH2 electrons → H+ gradient → ATP
Track the four stages of cellular respiration left-to-right on sketches: cytosol → matrix → inner membrane.
| Stage | Location | Major inputs | Major outputs |
|---|---|---|---|
| Glycolysis | Cytoplasm | Glucose, 2 ATP invested | 2 pyruvate, 4 ATP gross (net 2), 2 NADH |
| Pyruvate oxidation | Matrix | Pyruvate, NAD+, CoA | Acetyl-CoA, NADH, CO2 |
| Krebs cycle | Matrix | Acetyl-CoA | NADH, FADH2, small ATP/GTP, CO2 |
| ETC + chemiosmosis | Inner membrane | NADH, FADH2, O2 | Water, large ATP return |
Only glycolysis is oxygen-independent; downstream stages still need NAD+ recycling that ultimately depends on oxygen unless fermentation reroutes electrons.
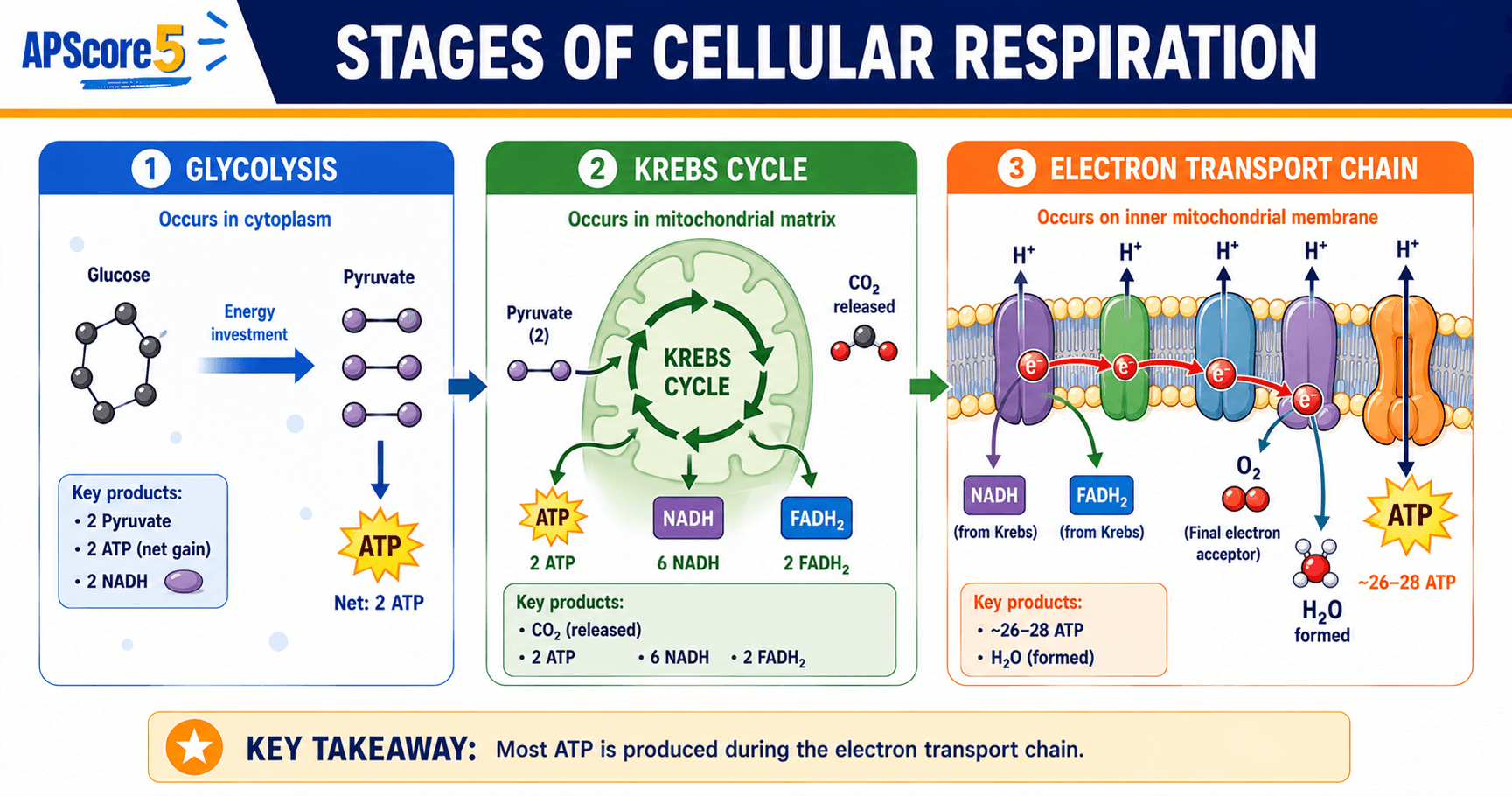
Stage 1: Glycolysis explained
Glycolysis AP Biology starts every aerobic story—memorize cytosol location before you touch mitochondria.
Glycolysis means “splitting sugar.” It is the first stage of cellular respiration and happens in the cytoplasm of every cell—plants, animals, fungi, and bacteria all run the same 10-step pathway.
AP trap: Glycolysis does not require oxygen, but aerobic respiration depends on oxygen because the ETC needs a final electron acceptor.
Inputs: one glucose (six carbons) plus two ATP molecules invested up front.
Outputs: two pyruvate (three carbons each), four ATP produced by substrate-level phosphorylation, and two NADH.
Net ATP: four ATP made minus two ATP invested equals net 2 ATP per glucose.
- Glycolysis is anaerobic—no oxygen is required for the reactions themselves.
- Memorize inputs and outputs; you rarely need intermediate enzyme names beyond AP lab prompts.
- Pyruvate enters mitochondria when oxygen is plentiful; otherwise it is diverted to fermentation.
- The two NADH molecules carry electrons that will eventually reach the electron transport chain.
- Because bacteria lack mitochondria but still perform glycolysis, the stage is evolutionarily ancient.
Glycolysis is the only stage that produces ATP strictly through substrate-level phosphorylation, where a phosphate group jumps directly from a metabolic intermediate onto ADP. The massive ATP return in stage 4 instead relies on chemiosmosis.
Stage 2: Pyruvate oxidation explained
Pyruvate oxidation AP Biology bridges cytosolic glycolysis to matrix Krebs—call it the link reaction when prompts show pyruvate crossing membranes.
After glycolysis, two pyruvate molecules cross the outer and inner mitochondrial membranes and arrive in the mitochondrial matrix. Each pyruvate is converted into acetyl-CoA.
Per pyruvate (multiply by two for one glucose): one carbon exits as CO2, NAD+ reduces to NADH, and the remaining two-carbon fragment bonds to coenzyme A.
Net per glucose: two acetyl-CoA, two NADH, and two CO2 molecules.
Teachers variously call this stage the link reaction or transition step; some textbooks bundle it with the Krebs cycle, others isolate it. AP questions still expect you to know its location and outputs.
The CO2 released here contributes to the breath you exhale—cellular respiration generates nearly all metabolic CO2 in animal tissues.
Stage 3: The Krebs cycle (citric acid cycle)
Krebs cycle AP Biology strips carbons as CO2 and loads NADH/FADH2—but it is not where most ATP appears.
The Krebs cycle (citric acid cycle or TCA cycle) spins in the mitochondrial matrix. Oxaloacetate combines with each incoming acetyl-CoA, and after eight enzymatic steps oxaloacetate re-forms—hence “cycle.”
Common mistake: Most ATP is not made in the Krebs cycle; most ATP comes from oxidative phosphorylation downstream.
Per acetyl-CoA (run twice per glucose): two carbons leave as CO2, three NAD+ reduce to NADH, FAD reduces to FADH2, and one ATP equivalent forms via substrate-level phosphorylation (GTP → ATP).
Per glucose (two turns): six NADH, two FADH2, about two ATP equivalents, and four CO2 molecules beyond what pyruvate oxidation already released.
By the time both turns finish, every carbon from the original glucose has been oxidized to CO2. The remaining useful energy now sits mostly in electron carriers, not in the carbon backbone.
Those carriers hand electrons forward to the electron transport chain, where oxygen will ultimately pull the electrons and enable massive ATP production.
Stage 4: Electron transport chain and oxidative phosphorylation
Electron transport chain AP Biology plus oxidative phosphorylation AP Biology captures most ATP—pair ETC chemistry with chemiosmosis cellular respiration language on every long answer.
The electron transport chain (ETC) is where most ATP forms. It resides on the inner mitochondrial membrane (including cristae) and partners with chemiosmosis.
Common mistake: Oxygen does not directly make ATP; it accepts electrons at the end of the ETC so the chain keeps moving.
AP trap: NADH and FADH2 are electron carriers, not ATP—they donate electrons that indirectly fuel ATP synthase.
- NADH and FADH2 donate electrons to the first complexes of the chain.
- Electrons travel through membrane complexes, releasing energy stepwise.
- That energy pumps H+ from the matrix into the intermembrane space, building an electrochemical gradient.
- Oxygen is the final electron acceptor, combining with H+ and electrons to produce water.
- H+ flows back through ATP synthase, converting gradient energy into ATP through oxidative phosphorylation.
ATP accounting shorthand: each NADH yields about 2.5 ATP, each FADH2 about 1.5 ATP, and combined they deliver roughly 26–28 ATP per glucose through chemiosmosis.
Without oxygen the chain jams: NADH cannot unload electrons, NAD+ pools shrink, pyruvate oxidation and the Krebs cycle stall, and the cell must ferment or die.
Oxygen final electron acceptor: O2 clears electrons from Complex IV—without it the proton pumps stall and chemiosmosis fails.
How does cellular respiration make ATP?
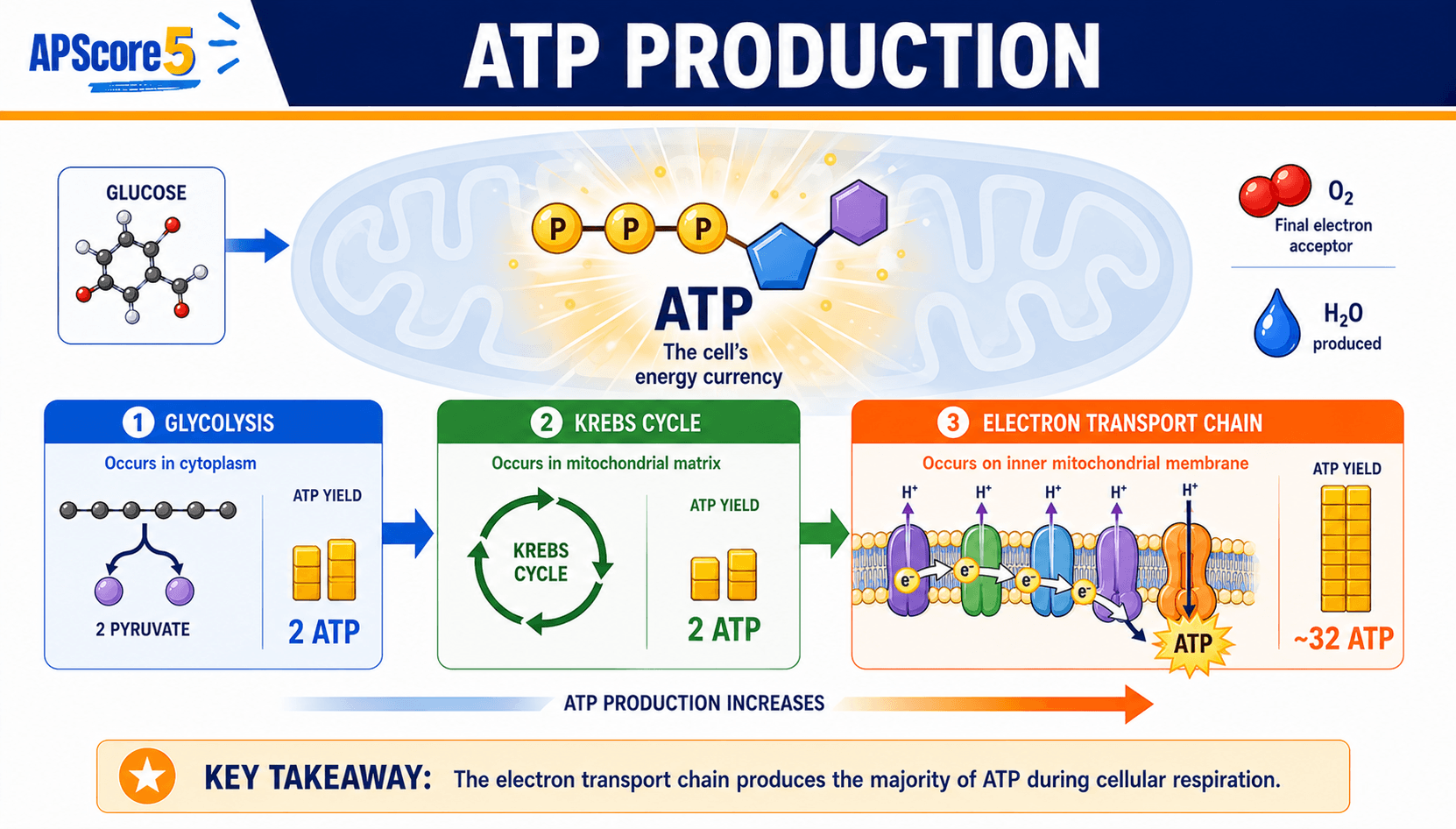
Key yields: glycolysis nets 2 ATP; Krebs contributes about 2 ATP per glucose via substrate-level steps; ETC/oxidative phosphorylation supplies about 26–28 ATP; total aerobic ≈ 30–32 ATP per glucose. Fermentation tops out near 2 ATP per glucose from glycolysis alone.
| Mechanism | Description | Typical share per glucose |
|---|---|---|
| Substrate-level phosphorylation | Phosphate transfer straight from intermediates to ADP | About 4 ATP |
| Oxidative phosphorylation | Proton motive force through ATP synthase | About 26–28 ATP |
| Total | Combined aerobic tally | About 30–32 ATP |
AP Bio note: Use ~30–32 ATP as the modern aerobic yield. Older textbooks may say 36–38, but AP questions usually care more about the source of ATP than the exact integer.
Key exam insight: Most ATP comes from oxidative phosphorylation tied to the proton gradient; only a minority arrives via substrate-level events.
Why ranges like 30–32 appear: Textbooks disagree about shuttle costs, membrane leakiness, and proton-to-ATP stoichiometry. The College Board expects conceptual totals near 30–32 ATP per glucose, not a single universal integer.
Aerobic vs anaerobic respiration
This aerobic vs anaerobic respiration table compares oxygen use, ATP yield cellular respiration totals, and fermentation end products.
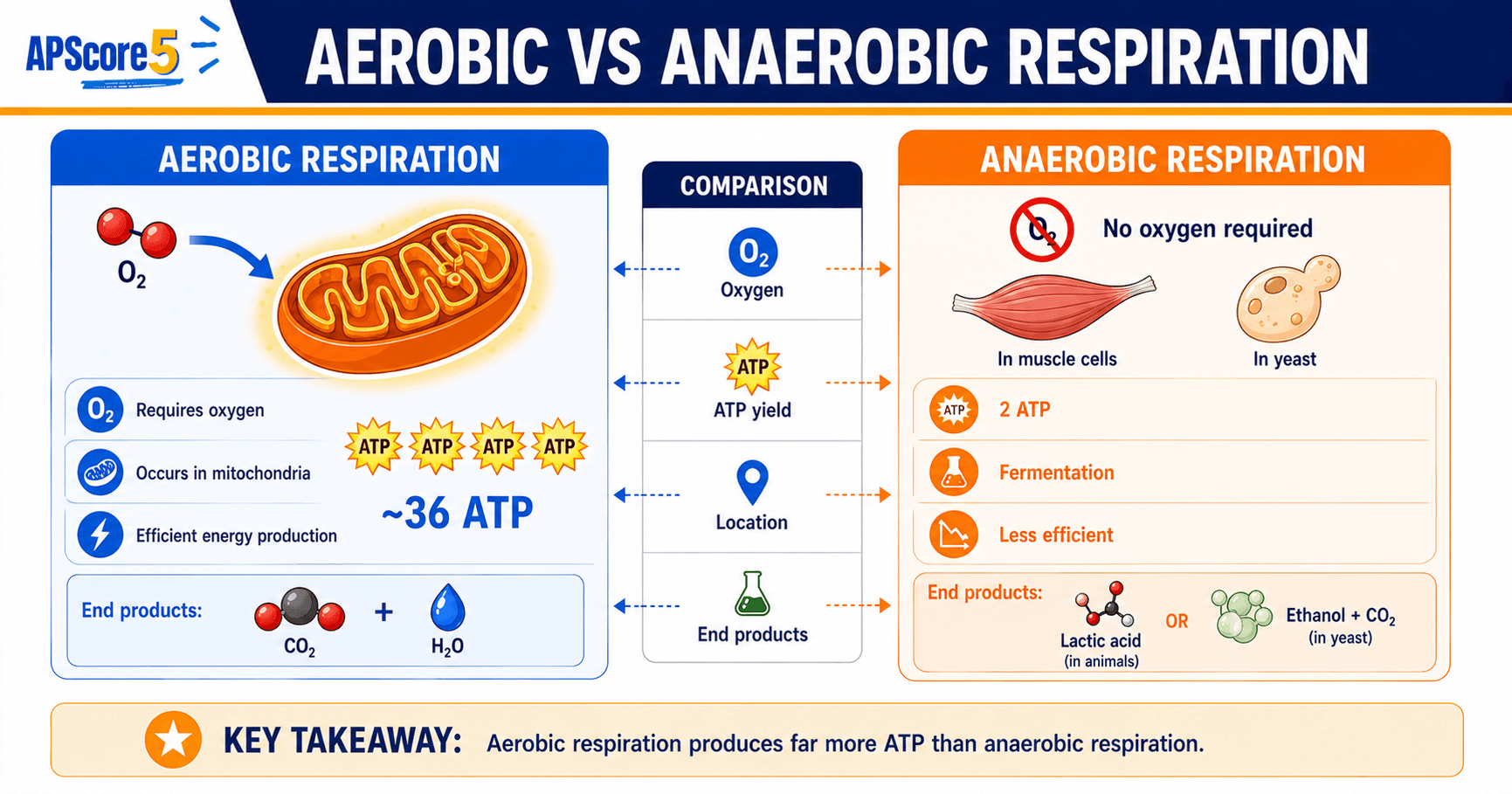
| Feature | Aerobic | Fermentation / anaerobic |
|---|---|---|
| Oxygen | Required for full pathway | Not required |
| ATP yield | ~30–32 per glucose | About 2 from glycolysis |
| End products | CO2 + H2O | Lactate or ethanol + CO2 |
| Purpose of fermentation | — | Regenerate NAD+ for glycolysis |
Sprint muscles experience lactate buildup when oxygen delivery lags behind ATP demand; yeast performing alcoholic fermentation illustrate the same NAD+ rescue logic.
Lactic acid fermentation converts pyruvate to lactate in hypoxic muscle.
Alcoholic fermentation converts pyruvate to ethanol plus CO2 in yeast—critical for bread leavening and brewing—and still tops out near two ATP per glucose because fermentation does not replace chemiosmosis.
Where cellular respiration shows up in living systems
Cardiac muscle relies on steady aerobic respiration; plentiful mitochondria keep contraction fueled during rest and sustained cardio.
Sprinting skeletal muscle temporarily tilts toward fermentation when oxygen cannot arrive fast enough—rapid glycolysis plus lactate regeneration preserves ATP turnover despite lower yield.
Brewer’s yeast in oxygen-rich starter cultures respire glucose fully; sealed fermenters force ethanol fermentation when oxygen drops, demonstrating how the same cell switches pathways.
Neurons spend ATP constantly on ion gradients; mitochondrial disorders that cripple the ETC therefore produce neurologic symptoms quickly.
Plant roots respire carbohydrates shipped from leaves at night; photosynthesis stores carbon and oxygen during the day, while mitochondria in every tissue burn sugars whenever metabolism demands ATP.
How cellular respiration appears on the AP Biology exam
This topic routinely contributes multiple multiple-choice items plus FRQ prompts that blend structure, data, and mechanism.
AP Exam Answer Template
Cellular respiration transfers energy from glucose to electron carriers, then uses the electron transport chain to pump H+ across the inner mitochondrial membrane. ATP synthase uses the proton gradient to make ATP, and oxygen keeps the chain moving by acting as the final electron acceptor.
In multiple-choice questions
- Krebs location: mitochondrial matrix—not inner membrane, not cytoplasm.
- Largest ATP yield: electron transport chain plus oxidative phosphorylation (chemiosmosis), not glycolysis.
- Final electron acceptor: molecular oxygen at Complex IV.
- Anoxic consequences: ETC stalls, NAD+ scarcity halts upstream oxidation unless fermentation recycles carriers.
- Oxygen-independent stage: glycolysis (fermentation also lacks oxygen but is separate vocabulary).
In free-response questions
- Predict effects of poisons that block ATP synthase or ETC complexes using proton-gradient logic.
- Interpret respirometry data showing O2 consumed versus CO2 released under varied substrates.
- Compare cellular respiration with photosynthesis overview at a glance before you mix up chemiosmosis direction.
- Trace electrons from NADH to oxygen and justify each energy-transducing step.
- Explain graphs of fermentation versus aerobic growth rates.
Common stimulus types
- Mitochondrion diagrams labeling matrix, intermembrane space, and cristae.
- Bar charts of ATP yield per stage.
- Tables of oxygen uptake under inhibitors.
- Schematics of ETC complexes with H+ pumping arrows.
AP writing formula: identify the stage or process → give location plus inputs/outputs → explain whether ATP forms via substrate-level phosphorylation or oxidative phosphorylation → connect to mitochondrial structure, oxygen’s role, or whole-cell energy budgets.
Test yourself in 5 seconds
A scientist adds a chemical that blocks ATP synthase in mitochondria. Which of the following is the first consequence?
Cellular respiration vs photosynthesis comparison
This cellular respiration vs photosynthesis comparison separates energy release (oxidation) from energy storage (light-driven carbon fixation).
| Feature | Cellular respiration | Photosynthesis |
|---|---|---|
| Overall reaction | C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + ATP | 6 CO2 + 6 H2O + light → C6H12O6 + 6 O2 |
| Purpose | Catabolic oxidation for ATP | Anabolic carbon fixation using light energy |
| Primary compartments | Cytoplasm + mitochondria | Chloroplasts (plants, algae, some bacteria) |
| Gas roles | Consumes O2; releases CO2 | Consumes CO2; releases O2 |
| Electron transport location | Inner mitochondrial membrane | Thylakoid membrane |
| ATP synthase | Driven by chemiosmosis during respiration | Driven by photophosphorylation chemiosmosis |
| Organisms | Nearly all eukaryotes + many prokaryotes | Photoautotrophs and some bacteria |
Ecologically, photosynthesis stores energy and releases oxygen while cellular respiration oxidizes fuels and returns CO2 for the Calvin cycle—two halves of the biosphere’s carbon and oxygen loop.
Both pathways reuse the chemiosmosis trick: electron transport generates an H+ gradient, and ATP synthase converts gradient potential into ATP.
Common AP Bio mistakes about cellular respiration
Score booster: Always include location, inputs, outputs, and mechanism when explaining a stage.
- Mixing locations: glycolysis stays in the cytoplasm; pyruvate oxidation and Krebs stay in the matrix; oxidative phosphorylation sits on inner membrane cristae.
- Claiming glycolysis needs oxygen: only downstream electron transport directly depends on O2 as terminal acceptor.
- Confusing ETC chemistry with ATP synthase: complexes pump protons; ATP synthase harvests the gradient.
- Oxidation vs reduction: NAD+ → NADH is reduction (gains electrons); NADH → NAD+ at the chain is oxidation—remember OIL RIG.
- Substrate-level vs oxidative phosphorylation: small ATP totals versus gradient-driven bulk synthesis.
- Equating lung ventilation with cellular respiration: breathing exchanges gases; mitochondria perform chemistry.
- Forgetting O2 as final acceptor: without it the chain stalls and NAD+ recycling fails unless fermentation intervenes.
How respirometry experiments connect to each stage
This respirometry AP Biology section connects oxygen consumption and carbon dioxide production to pathway activity—use it to justify which stage a drug or substrate perturbs.
Investigators often measure oxygen consumption or carbon dioxide production while manipulating substrates, inhibitors, or temperature. AP passages expect you to translate those measurements back into specific pathway steps.
Oxygen uptake rises when tissues burn fuel aerobically because the electron transport chain consumes O2 at Complex IV. If a poison blocks earlier complexes, O2 consumption may fall even though glucose is abundant because electrons never reach oxygen.
Carbon dioxide release tracks decarboxylation in pyruvate oxidation and the Krebs cycle. Adding malonate analogs that inhibit succinate dehydrogenase, for example, can stall the cycle and change CO2 timing even though glycolysis still runs.
Respiratory quotient (RQ) compares CO2 produced to O2 consumed. Pure carbohydrate oxidation approaches an RQ near 1.0, whereas fatty-acid breakdown yields proportionally less CO2 per O2, lowering RQ. Exam writers sometimes embed those ratios to check whether students understand that not every fuel follows glucose stoichiometry.
Uncoupler traces show elevated oxygen use without matching ATP gain because protons re-enter the matrix through alternative routes, bypassing ATP synthase. Students should explain that heat output rises while phosphorylation efficiency drops.
Rotenone or cyanide stories test whether you know where electrons accumulate. Rotenone near Complex I backs up NADH; cyanide at Complex IV stalls the entire chain. Either scenario eventually stops oxidative phosphorylation even if glycolysis limps along.
Yeast sugar switches remind you that fermentation replaces oxidative pathways when oxygen limits NAD+ recycling. Growth curves may slow dramatically after oxygen depletion even though glucose remains—because energy yield per glucose collapses.
When you rehearse those scenarios aloud, you rehearse the same evidence AP readers expect in paragraph proofs: name the measurement, connect it to the disturbed step, and state how NAD+/NADH balance responds.
Coupling those graphs with balanced reaction summaries keeps answers anchored: CO2 spikes usually mean decarboxylation somewhere between pyruvate oxidation and the Krebs cycle, whereas O2 dips trace electron movement toward oxygen. Separating the two signals prevents the classic mistake of blaming “respiration” generically without naming the stage.
Experimental repeats also matter: mitochondria isolated in buffer may show lag phases while substrates diffuse, so steady-state rates—not instant snapshots—support conclusions about inhibitor potency.
Extended reasoning drills for cellular respiration
Poisons and inhibitors
Complex blockers
Cyanide relatives stall Complex IV; electrons pile up and gradients collapse once pumps stop. Essays should mention NADH buildup and eventual pathway shutdown.
Uncouplers
DNP-like molecules dissipate proton gradients—oxygen consumption may surge while ATP synthesis falls because synthase lacks motive force.
Data interpretation
Respirometry graphs
Rising O2 consumption signals ETC demand; CO2 spikes trace pyruvate oxidation and Krebs releases. Pair graphs with temperature controls.
Yeast experiments
Anaerobic flasks produce ethanol plus dissolved CO2; aerobic cultures show fuller ATP budgets and biomass gain.
Writing discipline
Precise verbs
Use oxidized, reduced, pumped, coupled—avoid vague “happens” statements graders cannot reward.
Energy storytelling
Link glucose bond energy to NADH to proton gradients to ATP synthase torque so readers see the full transformation chain.
Structure drill: sketch a mitochondrion and label where glycolysis products enter, where CO2 exits, where O2 is consumed, and where ATP synthase spins—then narrate the pathway aloud.
Cellular respiration flashcards
Every fifth card advance triggers an ad placeholder with a three-second countdown before the next card appears.
Cellular respiration practice problems & AP Biology MCQs
Cellular respiration FRQ practice
Prompt: Yeast growing aerobically on glucose are treated with a chemical that blocks H+ flow through ATP synthase.
- (A) Identify where ATP synthase operates and describe its role.
- (B) Predict how ATP production changes and justify quantitatively.
- (C) Predict changes to proton distribution across the inner mitochondrial membrane.
- (D) Predict long-term survival with justification referencing fermentation.
Sample 4-point response
(A) ATP synthase operates during the electron transport chain / oxidative phosphorylation stage on the inner mitochondrial membrane. It allows H+ to flow from the intermembrane space back into the matrix, and that exergonic flow phosphorylates ADP—chemiosmosis.
(B) ATP production falls sharply because about 26–28 of the ~30–32 ATP per glucose normally come from oxidative phosphorylation. Blocking synthase leaves only the small ATP totals from substrate-level steps in glycolysis and the Krebs cycle—roughly a 90% drop in yield.
(C) H+ continues to be pumped into the intermembrane space while synthase is blocked, so the gradient steepens (high H+ outside the matrix, relatively lower inside) until the electron transport complexes can no longer pump efficiently and the chain slows.
(D) Yeast could survive briefly via alcoholic fermentation, but fermentation still nets only ~2 ATP per glucose—far below aerobic demand—so long-term growth, division, and biosynthesis would fail even though NAD+ recycling keeps glycolysis limping along.
Rubric (4 pts): (A) names oxidative phosphorylation and explains synthase role · (B) predicts dramatic ATP loss with quantitative reasoning · (C) describes proton buildup and gradient consequences · (D) contrasts short fermentation rescue with long-term energy deficit.
Cellular respiration FAQ
What is cellular respiration?
Cells oxidize glucose (and other fuels) to rebuild ATP. Aerobic cellular respiration runs glycolysis, pyruvate oxidation, the Krebs cycle, and the electron transport chain—oxygen pulls electrons through the chain so chemiosmosis can yield roughly 30–32 ATP per glucose.
What are the four stages of cellular respiration?
Glycolysis, pyruvate oxidation, the Krebs cycle, and the electron transport chain with oxidative phosphorylation—in that order for one glucose molecule.
Where does glycolysis happen?
In the cytoplasm (cytosol). No mitochondria are required for glycolysis itself.
Where does the Krebs cycle happen?
In the mitochondrial matrix (alongside pyruvate oxidation).
Where does the electron transport chain happen?
On the inner mitochondrial membrane (including cristae), embedded with ATP synthase.
What is the role of oxygen in cellular respiration?
Oxygen is the final electron acceptor at the end of the ETC. It receives depleted electrons and combines with H+ to form water, which keeps NAD+ recycling and the proton pumps running.
How much ATP does cellular respiration make?
Typically about 30–32 ATP per glucose aerobically—roughly 2 net from glycolysis, ~2 substrate-level from the Krebs portion, and ~26–28 from oxidative phosphorylation. Fermentation nets only ~2 ATP per glucose from glycolysis.
What is the difference between aerobic and anaerobic respiration?
Aerobic pathways use oxygen and complete oxidation through the ETC for high yield. Anaerobic fermentation skips oxygen-dependent ETC chemistry and regenerates NAD+ using fermentation products so glycolysis can continue at low yield.
What is chemiosmosis?
H+ pumped by the ETC creates a gradient across the inner membrane; when H+ flows through ATP synthase, ADP is phosphorylated—this couples electron transport to ATP synthesis.
What is oxidative phosphorylation?
ATP synthesis driven by the proton-motive force from the ETC (distinct from substrate-level phosphorylation in glycolysis/Krebs).
How does cellular respiration make ATP?
Substrate-level phosphorylation moves phosphate straight onto ADP in glycolysis and the Krebs cycle (~4 ATP equivalents per glucose). Oxidative phosphorylation uses chemiosmosis for the bulk (~26–28 ATP per glucose).
How is cellular respiration different from photosynthesis?
Respiration oxidizes glucose to extract ATP and releases CO2; photosynthesis reduces carbon using light energy and releases O2. Both reuse chemiosmosis but run opposite overall directions.
What does respirometry measure in AP Biology?
Typically oxygen consumed (linked to ETC activity) and/or carbon dioxide produced (linked to decarboxylation), sometimes respiratory quotient (CO2/O2) to infer substrate use.
What are NADH and FADH2?
Reduced electron carriers shuttling high-energy electrons from glycolysis, pyruvate oxidation, and the Krebs cycle to the ETC—they are not ATP themselves.